Niflumic acid
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| ATC code | |
| Pharmacokinetic data | |
| Elimination half-life | 2.5 hr[1] |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider |
|
| UNII | |
| KEGG |
|
| ChEMBL | |
| ECHA InfoCard |
100.022.289 |
| Chemical and physical data | |
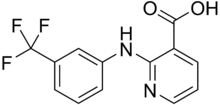
| Formula | C13H9F3N2O2 |
| Molar mass | 282.21797 g/mol |
| 3D model (JSmol) | |
| Melting point | 204 °C (399 °F) |
| |
| |
| | |
Niflumic acid is a drug used for joint and muscular pain. It is categorized as an inhibitor of cyclooxygenase-2. In experimental biology, it has been employed to inhibit chloride channels.[2] It has also been reported to act on GABA-A[3] and NMDA channels[4] and to block T-type calcium channels.[5]
References
- ↑ "Half life". Drug Bank. Retrieved 15 July 2011.
- ↑ Knauf, Philip A.; Mann, Nancy A (1984). "Use of niflumic acid to determine the nature of the asymmetry of the human erythrocyte anion exchange system". J. Gen. Physiol. 83: 703–725. doi:10.1085/jgp.83.5.703. PMC 2215658. PMID 6736917.
- ↑ Sinkkonen ST et al. (2003): Receptor subtype-dependent positive and negative modulation of GABA(A) receptor function by niflumic acid, a nonsteroidal anti-inflammatory drug, Mol Pharmacol, p. 753-63. PMID 12920213
- ↑ Lerma J., Martin d.R. (1992). "Chloride transport blockers prevent N-methyl-D-aspartate receptor-channel complex activation". Mol. Pharmacol. 41 (2): 217–222. PMID 1371581.
- ↑ Balderas E et al. (2012): Niflumic acid blocks native and recombinant T-type channels, J Cell Physiol, p. 2542-55. PMID 21898399
| Pyrazolones / Pyrazolidines | |
|---|---|
| Salicylates | |
| Acetic acid derivatives and related substances | |
| Oxicams | |
| Propionic acid derivatives (profens) |
|
| N-Arylanthranilic acids (fenamates) | |
| Coxibs | |
| Other | |
Items listed in bold indicate initially developed compounds of specific groups. #WHO-EM †Withdrawn drugs. ‡Veterinary use medications. | |
| Anti-inflammatory preparations, non-steroids |
| ||||||
|---|---|---|---|---|---|---|---|
| Capsaicin derivatives | |||||||
| Other | |||||||
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.