Funapide
 | |
| Clinical data | |
|---|---|
| Routes of administration | By mouth, topical |
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
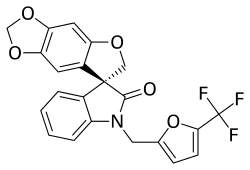
| Formula | C22H14F3NO5 |
| Molar mass | 429.34547 g/mol |
| 3D model (JSmol) | |
| |
| |
Funapide (INN) (former developmental code names TV-45070 and XEN402) is a novel analgesic under development by Xenon Pharmaceuticals in partnership with Teva Pharmaceutical Industries for the treatment of a variety of chronic pain conditions, including osteoarthritis, neuropathic pain, postherpetic neuralgia, and erythromelalgia, as well as dental pain.[1][2][3][4] It acts as a small-molecule Nav1.7 and Nav1.8 voltage-gated sodium channel blocker.[1][2][4] Funapide is being evaluated in humans in both oral and topical formulations, and as of July 2014, has reached phase IIb clinical trials.[1][3]
See also
References
- 1 2 3 Bagal, Sharan K.; Chapman, Mark L.; Marron, Brian E.; Prime, Rebecca; Ian Storer, R.; Swain, Nigel A. (2014). "Recent progress in sodium channel modulators for pain". Bioorganic & Medicinal Chemistry Letters. 24 (16): 3690–9. doi:10.1016/j.bmcl.2014.06.038. ISSN 0960-894X. PMID 25060923.
- 1 2 Stephen McMahon; Martin Koltzenburg; Irene Tracey; Dennis C. Turk (1 March 2013). Wall & Melzack's Textbook of Pain: Expert Consult - Online. Elsevier Health Sciences. p. 508. ISBN 0-7020-5374-0.
- 1 2 Xenon Pharma. "TV-45070: A Small Molecule for the Treatment of the Orphan Disease EM and Other Pain Disorders".
- 1 2 Xenon Pharma (2012). "Teva and Xenon Announce Teva's World Wide License of Xenon's Pain Drug XEN402".
External links
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.