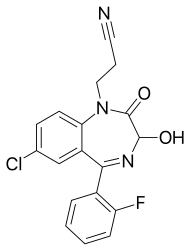
Cinolazepam
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Gerodorm |
| AHFS/Drugs.com | cinolazepam |
| Routes of administration | Oral |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 90–100% |
| Metabolism | Hepatic |
| Elimination half-life | 9 hours[1] |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C18H13ClFN3O2 |
| Molar mass | 357.8 |
| 3D model (JSmol) | |
| Chirality | Racemic mixture |
| |
| |
| | |
Cinolazepam[2] (marketed under the brand name Gerodorm)[3] is a drug which is a benzodiazepine derivative. It possesses anxiolytic, anticonvulsant, sedative and skeletal muscle relaxant properties. Due to its strong sedative properties, it is primarily used as a hypnotic.
Cinolazepam is not approved for sale in the United States or Canada.
See also
References
- ↑ Saletu, B.; G. Kindshofer; P. Anderer; J. Grunberger (1987). "Short-term sleep laboratory studies with cinolazepam in situational insomnia induced by traffic noise". International Journal of Clinical Pharmacology Research. 7 (5): 407&ndash, 18. PMID 2889679.
- ↑ US Patent 4388313 Novel 3-hydroxy-1,4-benzodiazepine-2-ones and process for the preparation thereof
- ↑ Lannacher Romania (1999). "Gerodorm". Produse Gerot inregistrate in Romania (in Romanian). Archived from the original on 11 October 2006. Retrieved 17 August 2006.
External links
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.