Tribromoethanol
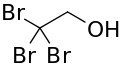 | |
| Clinical data | |
|---|---|
| Trade names | Avertin |
| Synonyms | Tribromoethyl alcohol |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C2H3Br3O |
| Molar mass | 282.76 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 73–79 °C (163–174 °F) [1] |
| Boiling point | 92–93 °C (198–199 °F) at 10 mmHg[1] |
| |
| |
Tribromoethanol is a sedative. It is used to anesthetize laboratory animals, particularly rodents, prior to surgery.[2] As a solution in tert-amyl alcohol, it has the brand name Avertin.[3] Tribromoethanol causes rapid and deep anesthesia followed by rapid and full postoperative recovery.[4]
Tribromoethanol was widely used as a general anesthetic in humans in the first half of the twentieth century.[5] Electrophysiology studies showed that tribromoethanol acts as a positive allosteric modulator of the inhibitory GABAA and glycine receptors, a mechanism similar to that seen with the related compound 2,2,2-trichloroethanol.[6] Bromal hydrate (2,2,2-tribromoethanol-1,1-diol), a compound also recognized to produce general anesthesia in animals, is metabolized to tribromoethanol.[7]
See also
References
- 1 2 "2,2,2-Tribromoethanol". Sigma-Aldrich.
- ↑ "Tribromoethanol (Avertin)". Cold Spring Harbor Protocols. Cold Spring Harbor Laboratory.
- ↑ "Guidelines for the Use of Tribromoethanol/Avertin Anesthesia" (PDF). National Cancer Institute.
- ↑ "Tribromoethanol (Avertin) as an anaesthetic in mice" (PDF). Laboratory Animals. 33: 192–193. 1999.
- ↑ G Edwards (1945). "Tribromethyl alcohol (avertin, bromethol), 1928-1945". Proceedings of the Royal Society of Medicine. 39: 71–76. PMID 21010258.
- ↑ M.D. Krasowski & N.L. Harrison (2000). "The actions of ether, alcohol and alkane general anaesthetics on GABAA and glycine receptors and the effects of TM2 and TM3 mutations". British Journal of Pharmacology. 129 (4): 731–743. doi:10.1038/sj.bjp.0703087. PMC 1571881. PMID 10683198.
- ↑ Lehman G and Knoefel PK (1938). Trichloroethanol, tribromoethanol, chloral hydrate, and bromal hydrate. J Pharmacol Exp Ther, 63: 453-465.