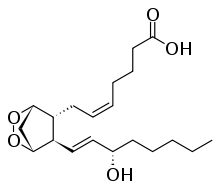
Prostaglandin H2
 | |
| Names | |
|---|---|
| Other names
PGH2, Endoperoxide H2, Prostaglandin R2 | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| MeSH | Prostaglandin+H2 |
PubChem CID |
|
| |
| |
| Properties | |
| C20H32O5 | |
| Molar mass | 352.465 g/mol |
| Density | 1.129 ± 40.0 g/mL |
| Boiling point | 490 ± 40.0 °C |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Prostaglandin H2 is a type of prostaglandin and a precursor for many other biologically significant molecules. It is synthesized from arachidonic acid in a reaction catalyzed by a cyclooxygenase enzyme.[1]

Eicosanoid synthesis - prostaglandin H2 near center
It is acted upon by:
- Prostacyclin synthase to create prostacyclin
- Thromboxane-A synthase to create thromboxane A2 and 12-(S)-hydroxy-5Z,8E,10E-heptadecatrienoic acid (HHT) (see 12-Hydroxyheptadecatrienoic acid)
- Prostaglandin D2 synthase to create prostaglandin D2
- Prostaglandin E synthase to create prostaglandin E2
It rearranges non-enzymatically to:
- A mixture of 12-(S)-hydroxy-5Z,8E,10E-heptadecatrienoic acid (HHT) and 12-(S)-hydroxy-5Z,8Z,10E-heptadecatrienoic acid (see 12-Hydroxyheptadecatrienoic acid)
Use of Prostaglandin H2:
- regulating the constriction and dilation of blood vessels
- stimulating platelet aggregation
Effects of Aspirin on Prostaglandin H2:
- Aspirin has been hypothesized to block the conversion of arachidonic acid to Prostaglandin
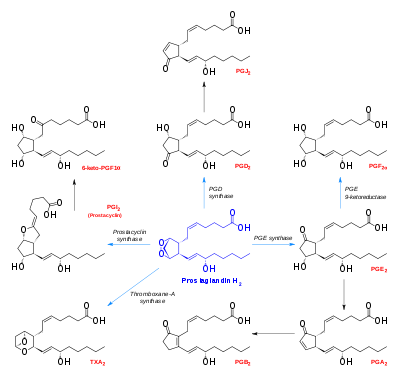
Figure 1: Synthetic pathways from PGH2 (the parent compound) to prostaglandins, prostacyclin and thromboxanes
References
- ↑ "The Cyclooxygenase Reaction Mechanism". ACS Publications. Retrieved 18 April 2016.
2. "Prostaglandin H2". SciFinder. Retrieved 22 October 2017.
3. "Acetylation of Prostaglandin H2 Synthases by Aspirin is Inhibited by Redox Cycling of the Peroxidase". "Biochem Pharmacol. Retrieved 22 October 2017.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.