Testosterone undecanoate
 | |
 | |
| Clinical data | |
|---|---|
| Pronunciation |
/tɛˈstɒstəroʊn |
| Trade names | Nebido, Aveed, Andriol, others |
| Synonyms | TU; Testosterone undecylate; Testosterone 17β-undecanoate; ORG-538; CLR-610 |
| Pregnancy category |
|
| Routes of administration | By mouth, intramuscular injection |
| Drug class | Androgen; Anabolic steroid; Androgen ester |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability |
Oral: 3–7% Intramuscular: high |
| Protein binding | High (testosterone) |
| Metabolism | Liver |
| Metabolites | Testosterone, undecanoic acid, metabolites of testosterone |
| Elimination half-life |
In TSO: 20.9 days (i.m.)[1][2] In CO: 33.9 days (i.m.)[1][2] |
| Excretion | Urine |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard |
100.025.193 |
| Chemical and physical data | |
| Formula | C30H48O3 |
| Molar mass | 456.711 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Testosterone undecanoate, sold under the brand names Nebido, Aveed, and Andriol among others, is an androgen and anabolic steroid (AAS) medication which is used mainly in the treatment of low testosterone levels in men.[3][1][4][5] It is also used in hormone therapy for transgender men.[6][7][8] It is taken by mouth two to three times per day with food or given by injection into muscle once every 12 weeks or so.[5][9]
Side effects of testosterone undecanoate include symptoms of masculinization like acne, increased hair growth, voice changes, and increased sexual desire.[5] The drug is a synthetic androgen and anabolic steroid and hence is an agonist of the androgen receptor (AR), the biological target of androgens like testosterone and dihydrotestosterone (DHT).[10][5] It has strong androgenic effects and moderate anabolic effects, which make it useful for producing masculinization and suitable for androgen replacement therapy.[5] Testosterone undecanoate is a testosterone ester and a prodrug of testosterone in the body.[4][3][1] Because of this, it is considered to be a natural and bioidentical form of testosterone.[11]
Testosterone undecanoate was introduced in China for use by injection and in Europe for use by mouth in the 1970s.[12][13] It became available for use by injection in Europe in the early to mid 2000s and in the United States in 2014.[14][15] A formulation for use by mouth is not currently available in the United States but is pending approval as of 2018.[16] Along with testosterone enanthate, testosterone cypionate, and testosterone propionate, testosterone undecanoate is one of the most widely used testosterone esters.[10][1][5] However, it has advantages over other testosterone esters in that it can be taken by mouth and in that it has a far longer duration when given by injection.[17][3][1][2][5] In addition to its medical use, testosterone undecanoate is used to improve physique and performance.[5] The drug is a controlled substance in many countries and so non-medical use is generally illicit.[5]
Medical uses
Testosterone undecanoate is used in androgen replacement therapy. It is specifically approved only for the treatment of hypogonadism.[18][19][20] As an intramuscular injection, it is administered at a dosage of 1,000 mg once every 12 weeks.[9] Conversely, oral testosterone undecanoate must be taken two or three times a day with food.[9][21]
Side effects
Side effects of testosterone undecanoate include virilization among others.[5]
Anaphylaxis
The Reandron 1000 formulation (Nebido in the United States) contains 1,000 mg of testosterone undecanoate suspended in castor oil with benzyl benzoate for solubilization and as a preservative, and is administered by intramuscular injection. As an excipient, benzyl benzoate has been reported as a cause of anaphylaxis in a case in Australia.[22] Bayer includes this report in information for health professionals and recommends that physicians "should be aware of the potential for serious allergic reactions" to preparations of this type.[23] In Australia, reports to ADRAC, which evaluates reports of adverse drug reactions for the Therapeutic Goods Administration, show several reports of allergic issues since the anaphylaxis case from 2011.
Pharmacology
Pharmacodynamics
Testosterone undecanoate is a prodrug of testosterone and is an androgen and anabolic–androgenic steroid (AAS). That is, it is an agonist of the androgen receptor (AR).
Pharmacokinetics
Testosterone undecanoate has a very long elimination half-life and mean residence time when given as a depot intramuscular injection.[24][1][2] Its elimination half-life is 20.9 days and its mean residence time is 34.9 days in tea seed oil, while its elimination half-life is 33.9 days and its mean residence time is 36.0 days in castor oil.[1][2] These values are substantially longer than those of testosterone enanthate (which, in castor oil, has values of 4.5 days and 8.5 days, respectively).[24] Testosterone undecanoate is administered via intramuscular injection once every three months or so.[9][25]
Chemistry
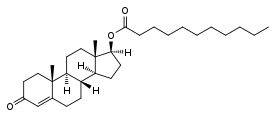
Testosterone undecanoate, or testosterone 17β-undecanoate, is a synthetic androstane steroid and a derivative of testosterone.[26][27] It is an androgen ester; specifically, it is the C17β undecylate (undecanoate) ester of testosterone.[26][27] A related testosterone ester with a similarly very long duration is testosterone buciclate.[3][4]
History
In the late 1970s, testosterone undecanoate was introduced for oral use in Europe,[12] although intramuscular testosterone undecanoate had already been in use in China for several years.[13] Intramuscular testosterone undecanoate was not introduced in Europe and the United States until much later, in the early to mid 2000s and 2014, respectively.[14][15] Testosterone undecanoate was approved in the United States after three previous rejections due to safety concerns.[28]
Society and culture
Generic names
Testosterone undecanoate is the generic name of the drug and its USAN and BAN.[26][27][29][30] It is also referred to as testosterone undecylate.[26][27][29][30]
Brand names
Testosterone undecanoate is or has been marketed under a variety of brand names including Aveed, Andriol, Androxon, Cernos Depot, Nebido, Panteston, Restandol, Nebido-R, Reandron 1000, and Undestor.[26][27][29][30]
Availability
Intramuscular testosterone undecanoate is available in the United States, Canada, Europe, and elsewhere in the world.[5][31][32] It is approved in over 100 countries worldwide.[31][5] Oral testosterone undecanoate is available in Canada, Europe, Mexico, Asia, and elsewhere but not in the United States.[31][33] Intramuscular testosterone undecanoate is marketed most commonly as Nebido in Canada and Europe and as Aveed in the United States while oral testosterone undecanoate is marketed most commonly as Andriol.[5][31][32]
Legal status
Testosterone undecanoate, along with other AAS, is a schedule III controlled substance in the United States under the Controlled Substances Act and a schedule IV controlled substance in Canada under the Controlled Drugs and Substances Act.[34][35]
Research
As of June 2017, an oral formulation of testosterone undecanoate is in preregistration for the treatment of hypogonadism in the United States.[16]
As of January 2017, intramuscular testosterone undecanoate is in phase II clinical trials for the treatment of non-alcoholic steatohepatitis in the United Kingdom.[18]
References
- 1 2 3 4 5 6 7 8 Eberhard Nieschlag; Hermann M. Behre; Susan Nieschlag (13 January 2010). Andrology: Male Reproductive Health and Dysfunction. Springer Science & Business Media. pp. 441–446. ISBN 978-3-540-78355-8.
- 1 2 3 4 5 Behre HM, Abshagen K, Oettel M, Hübler D, Nieschlag E (1999). "Intramuscular injection of testosterone undecanoate for the treatment of male hypogonadism: phase I studies". Eur. J. Endocrinol. 140 (5): 414–9. doi:10.1530/eje.0.1400414. PMID 10229906.
- 1 2 3 4 Eberhard Nieschlag; Hermann M. Behre; Susan Nieschlag (26 July 2012). Testosterone: Action, Deficiency, Substitution. Cambridge University Press. pp. 313–315, 321–322. ISBN 978-1-107-01290-5.
- 1 2 3 Kenneth L. Becker (2001). Principles and Practice of Endocrinology and Metabolism. Lippincott Williams & Wilkins. pp. 1185–. ISBN 978-0-7817-1750-2.
- 1 2 3 4 5 6 7 8 9 10 11 12 13 William Llewellyn (2011). Anabolics. Molecular Nutrition Llc. pp. 180–182, 331–334. ISBN 978-0-9828280-1-4.
- ↑ Irwig MS (2017). "Testosterone therapy for transgender men". Lancet Diabetes Endocrinol. 5 (4): 301–311. doi:10.1016/S2213-8587(16)00036-X. PMID 27084565.
- ↑ JW Jacobeit; LJ Gooren; HM Schulte (2007). "Long-acting intramuscular testosterone undecanoate for treatment of female-to-male transgender individuals". The Journal of Sexual Medicine. 4 (5): 1479–84. doi:10.1111/j.1743-6109.2007.00556.x. PMID 17635694.
- ↑ JW Jacobeit; LJ Gooren; HM Schulte (2009). "Safety aspects of 36 months of administration of long-acting intramuscular testosterone undecanoate for treatment of female-to-male transgender individuals". European Journal of Endocrinology. 161 (5): 795–8. doi:10.1530/EJE-09-0412. PMID 19749027.
- 1 2 3 4 S. Bertelloni; O. Hiort (28 September 2010). New Concepts for Human Disorders of Sexual Development. S. Karger AG. pp. 256–. ISBN 978-3-8055-9569-8.
- 1 2 Kicman AT (2008). "Pharmacology of anabolic steroids". Br. J. Pharmacol. 154 (3): 502–21. doi:10.1038/bjp.2008.165. PMC 2439524. PMID 18500378.
- ↑ Santoro N, Braunstein GD, Butts CL, Martin KA, McDermott M, Pinkerton JV (2016). "Compounded Bioidentical Hormones in Endocrinology Practice: An Endocrine Society Scientific Statement". J. Clin. Endocrinol. Metab. 101 (4): 1318–43. doi:10.1210/jc.2016-1271. PMID 27032319.
- 1 2 Hoberman J (21 February 2005). Testosterone Dreams: Rejuvenation, Aphrodisia, Doping. University of California Press. pp. 134–. ISBN 978-0-520-93978-3.
- 1 2 Mundy AR, Fitzpatrick J, Neal DE, George NJ (26 July 2010). The Scientific Basis of Urology. CRC Press. pp. 294–. ISBN 978-1-84184-749-8.
- 1 2 Melmed S, Polonsky KS, Larsen PR, Kronenberg HM (11 November 2015). Williams Textbook of Endocrinology. Elsevier Health Sciences. pp. 709, 711, 765. ISBN 978-0-323-34157-8.
- 1 2 Adis R&D Profile (2004). "Testosterone Undecanoate—Schering AG". Drugs. 5 (6): 368–369. doi:10.2165/00126839-200405060-00012.
- 1 2 http://adisinsight.springer.com/drugs/800027560
- ↑ Köhn, Frank-Michael; Schill, Wolf-Bernhard (November 2003). "A new oral testosterone undecanoate formulation". World Journal of Urology. 21 (5): 311–315. doi:10.1007/s00345-003-0372-x. PMID 14579074.
- 1 2 http://adisinsight.springer.com/drugs/800017041
- ↑ http://adisinsight.springer.com/drugs/800011489
- ↑ https://www.accessdata.fda.gov/drugsatfda_docs/label/2014/022219s000lbl.pdf
- ↑ Jean L. Fourcroy (27 October 2008). Pharmacology, Doping and Sports: A Scientific Guide for Athletes, Coaches, Physicians, Scientists and Administrators. Routledge. pp. 25–. ISBN 978-1-134-08880-5.
- ↑ Ong, G. S. Y.; Somerville, C. P.; Jones, T. W.; Walsh, J. P. (2012). "Anaphylaxis Triggered by Benzyl Benzoate in a Preparation of Depot Testosterone Undecanoate". Case Rep Med. 2012. doi:10.1155/2012/384054. PMC 3261473. PMID 22272209. 384054.
- ↑ "Nebido Monograph – Information for Health Care Professionals". Bayer. 2016. Retrieved 19 October 2016.
- 1 2 Anita H. Payne; Matthew P. Hardy (28 October 2007). The Leydig Cell in Health and Disease. Springer Science & Business Media. pp. 423–. ISBN 978-1-59745-453-7.
- ↑ Yeung SJ, Escalante CP, Gagel RF (2009). Medical Care of Cancer Patients. PMPH-USA. pp. 247–. ISBN 978-1-60795-008-0.
- 1 2 3 4 5 J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. pp. 641–642. ISBN 978-1-4757-2085-3.
- 1 2 3 4 5 Index Nominum 2000: International Drug Directory. Taylor & Francis. January 2000. pp. 1002–1004. ISBN 978-3-88763-075-1.
- ↑ Miriam E. Tucker (March 7, 2014). "FDA Approves Aveed Testosterone Jab, with Restrictions". Medscape. Retrieved December 13, 2016.
- 1 2 3 I.K. Morton; Judith M. Hall (6 December 2012). Concise Dictionary of Pharmacological Agents: Properties and Synonyms. Springer Science & Business Media. ISBN 978-94-011-4439-1.
- 1 2 3 https://www.drugs.com/international/testosterone.html
- 1 2 3 4 Nieschlag, Eberhard; Nieschlag, Susan (2017). "The History of Testosterone and The Testes: From Antiquity to Modern Times": 1–19. doi:10.1007/978-3-319-46086-4_1.
- 1 2 "Drug Product Database - Health Canada". Health Canada. Archived from the original on November 19, 2016. Retrieved November 13, 2016.
- ↑ Arthur P. Arnold; Donald W. Pfaff; Anne M. Etgen; Susan E. Fahrbach, Robert T. Rubin (10 June 2002). Hormones, Brain and Behavior, Five-Volume Set. Academic Press. pp. 20–. ISBN 978-0-12-532104-4.
- ↑ Steven B. Karch, MD, FFFLM (21 December 2006). Drug Abuse Handbook, Second Edition. CRC Press. pp. 30–. ISBN 978-1-4200-0346-8.
- ↑ Linda Lane Lilley; Julie S. Snyder; Shelly Rainforth Collins (5 August 2016). Pharmacology for Canadian Health Care Practice. Elsevier Health Sciences. pp. 50–. ISBN 978-1-77172-066-3.
External links
- Andriol (testosterone undecanoate) - William Llewellyn's Anabolic.org
- Testosterone undecanoate (intramuscular) - AdisInsight
- Testosterone undecanoate (oral) - AdisInsight