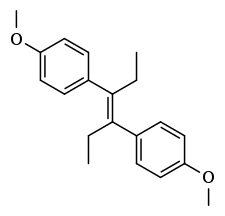
Dimestrol
 | |
| Clinical data | |
|---|---|
| Trade names | Depot-Ostromon; Synthila |
| Synonyms | Dianisylhexene; 4,4'-Dimethoxy-α,α'-diethylstilbene; Diethylstilbestrol dimethyl ether; Dimethoxydiethylstilbestrol; (E)-4,4'-(1,2-Diethylethylene)dianisole |
| Drug class | Nonsteroidal estrogen; Estrogen ether |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| Chemical and physical data | |
| Formula | C20H24O2 |
| Molar mass | 296.403 g/mol |
| 3D model (JSmol) | |
| |
| |
Dimestrol (brand names Depot-Ostromon, Synthila), also known as dianisylhexene, 4,4'-dimethoxy-α,α'-diethylstilbene, diethylstilbestrol dimethyl ether, and dimethoxydiethylstilbestrol, is a synthetic nonsteroidal estrogen of the stilbestrol group which is related to diethylstilbestrol.[1][2] It has been used to induce the development of female secondary sexual characteristics in the case of female delayed puberty or hypogonadism.[3] The drug has also been used as a fattening agent in livestock.[4]
See also
References
- ↑ J. Elks (14 November 2014). The Dictionary of Drugs: Chemical Data: Chemical Data, Structures and Bibliographies. Springer. p. 396. ISBN 978-1-4757-2085-3.
- ↑ William Andrew Publishing (22 October 2013). Pharmaceutical Manufacturing Encyclopedia, 3rd Edition. Elsevier. pp. 1324–. ISBN 978-0-8155-1856-3.
- ↑ Soviet Genetics. Consultants Bureau. 1982.
- ↑ National Research Council (U.S.). Committee on Animal Nutrition (1953). Hormonal Relationships and Applications in the Production of Meats, Milk, and Eggs: A Report of the Committee on Animal Nutrition. National Academies. pp. 5–13. NAP:14582.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.