Hypotaurine
 | |
 | |
| Names | |
|---|---|
| IUPAC name
2-Aminoethanesulfinic acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.155.825 |
| KEGG | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C2H7NO2S | |
| Molar mass | 109.15 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Hypotaurine is a sulfinic acid that is an intermediate in the biosynthesis of taurine. Like taurine, it also acts as an endogenous neurotransmitter via action on the glycine receptors.[1]
Hypotaurine is derived from cysteine (and homocysteine). In mammals, the biosynthesis of hypotaurine from cysteine occurs in the pancreas. In the cysteine sulfinic acid pathway, cysteine is first oxidized to its sulfinic acid, catalyzed by the enzyme cysteine dioxygenase. Cysteine sulfinic acid, in turn, is decarboxylated by sulfinoalanine decarboxylase to form hypotaurine. Hypotaurine is enzymatically oxidized to yield taurine by hypotaurine dehydrogenase.[2]
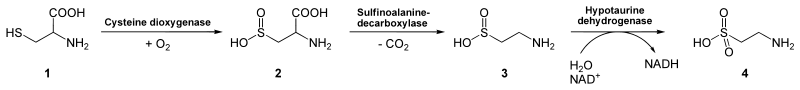
Hypotaurine (3) is an intermediate in the conversion of cysteine (1) to taurine (4).
References
- ↑ Kalir, Asher; Kalir, Henry H. "Biological activity of sulfinic acid derivatives" in Chemistry of Sulphinic Acids, Esters Their Derivatives Edited by Patai, Saul. Wiley, New York, 1990, pp. 665.
- ↑ Sumizu K (1962). "Oxidation of hypotaurine in rat liver". Biochim. Biophys. Acta. 63: 210–212. doi:10.1016/0006-3002(62)90357-8. PMID 13979247.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.