List of therapeutic monoclonal antibodies
This is a list of therapeutic, diagnostic and preventive monoclonal antibodies, antibodies that are clones of a single parent cell. When used as drugs, the International Nonproprietary Names (INNs) end in -mab. The remaining syllables of the INNs, as well as the column Source, are explained in Nomenclature of monoclonal antibodies.
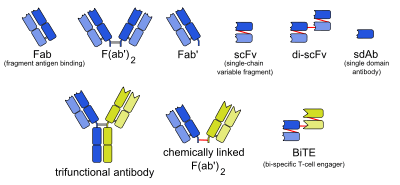
Types of monoclonal antibodies with other structures than naturally occurring antibodies.
The abbreviations in the column Type are as follows:
- mab: whole monoclonal antibody
- Fab: fragment, antigen-binding (one arm)
- F(ab')2: fragment, antigen-binding, including hinge region (both arms)
- Fab': fragment, antigen-binding, including hinge region (one arm)
- Variable fragments:
- scFv: single-chain variable fragment
- di-scFv: dimeric single-chain variable fragment
- sdAb: single-domain antibody
- Bispecific monoclonal antibodies:
- 3funct: trifunctional antibody
- BiTE: bi-specific T-cell engager
This list of over 500 monoclonal antibodies includes approved and investigational drugs as well as drugs that have been withdrawn from market; consequently, the column Use does not necessarily indicate clinical usage. See the list of FDA approved therapeutic monoclonal antibodies in the Monoclonal antibody therapy page.
| Name | Trade name | Type | Source | Target | Approved | Use |
|---|---|---|---|---|---|---|
| 3F8 | mab | mouse | GD2 ganglioside | neuroblastoma | ||
| 8H9[1] | mab | mouse | B7-H3 | neuroblastoma, sarcoma, metastatic brain cancers | ||
| Abagovomab[2] | mab | mouse | CA-125 (imitation) | ovarian cancer | ||
| Abciximab | ReoPro | Fab | chimeric | CD41 (integrin alpha-IIb) | Y | platelet aggregation inhibitor |
| Abituzumab[3] | mab | humanized | CD51 | cancer | ||
| Abrezekimab[4] | Fab | humanized | interleukin 13 | ? | ||
| Abrilumab | mab | human | integrin α4β7 | inflammatory bowel disease, ulcerative colitis, Crohn's disease | ||
| Actoxumab | mab | human | Clostridium difficile | Clostridium difficile colitis | ||
| Adalimumab | Humira | mab | human | TNF-α | Y | Rheumatoid arthritis, Crohn's disease, Plaque Psoriasis, Psoriatic Arthritis, Ankylosing Spondylitis, Juvenile Idiopathic Arthritis, Hemolytic disease of the newborn |
| Adecatumumab[5] | mab | human | EpCAM | prostate and breast cancer | ||
| Atidortoxumab[6] | mab | human | Staphylococcus aureus alpha toxin | ? | ||
| Aducanumab | mab | human | beta-amyloid | Alzheimer's disease | ||
| Afasevikumab[7] | mab | human | IL17A and IL17F | multiple sclerosis | ||
| Afelimomab | F(ab')2 | mouse | TNF-α | sepsis | ||
| Afutuzumab[8] | mab | humanized | CD20 | lymphoma | ||
| Alacizumab pegol[9] | F(ab')2 | humanized | VEGFR2 | cancer | ||
| Alemtuzumab[10] | Lemtrada, Campath | mab | humanized | CD52 | Y | Multiple sclerosis |
| Alirocumab | Praluent | mab | human | PCSK9 | Y | hypercholesterolemia |
| Altumomab pentetate | Hybri-ceaker | mab | mouse | CEA | colorectal cancer (diagnosis) | |
| Amatuximab | mab | chimeric | mesothelin | cancer | ||
| Anatumomab mafenatox | Fab | mouse | TAG-72 | non-small cell lung carcinoma | ||
| Andecaliximab | mab | chimeric | gelatinase B | gastric cancer or gastroesophageal junction adenocarcinoma | ||
| Anetumab ravtansine[3] | mab | human | MSLN | cancer | ||
| Anifrolumab | mab | human | interferon α/β receptor | systemic lupus erythematosus | ||
| Anrukinzumab[9] (= IMA-638)[11] | mab | humanized | IL-13 | asthma | ||
| Apolizumab[12] | mab | humanized | HLA-DR ? | hematological cancers | ||
| Aprutumab ixadotin[13] | mab | human | FGFR2 | ? | ||
| Arcitumomab | CEA-Scan | Fab' | mouse | CEA | gastrointestinal cancers (diagnosis) | |
| Ascrinvacumab | mab | human | activin receptor-like kinase 1 | cancer | ||
| Aselizumab[14] | mab | humanized | L-selectin (CD62L) | severely injured patients | ||
| Atezolizumab[15] | Tecentriq | mab | humanized | PD-L1 | Y | cancer |
| Atinumab[16] | mab | human | RTN4 | ? | ||
| Atorolimumab | mab | human | Rhesus factor | hemolytic disease of the newborn | ||
| Avelumab[7] | Bavencio | mab | human | PD-L1 | Y | cancer |
| Azintuxizumab vedotin[17] | mab | chimeric/humanized | CD319 | cancer | ||
| Bapineuzumab[18] | mab | humanized | beta amyloid | Alzheimer's disease | ||
| Basiliximab | Simulect | mab | chimeric | CD25 (α chain of IL-2 receptor) | Y | prevention of organ transplant rejections |
| Bavituximab[2] | mab | chimeric | phosphatidylserine | cancer, viral infections | ||
| BCD-100 | ? | human | PD-1 | melanoma | ||
| Bectumomab | LymphoScan | Fab' | mouse | CD22 | non-Hodgkin's lymphoma (detection) | |
| Begelomab[19] | mab | mouse | DPP4 | ? | ||
| Belantamab mafodotin[4] | mab | humanized | BCMA | cancer | ||
| Belimumab | Benlysta | mab | human | BAFF | Y | non-Hodgkin lymphoma etc. |
| Bemarituzumab[6] | mab | humanized | FGFR2 | gastric cancer or gastroesophageal junction adenocarcinoma | ||
| Benralizumab | Fasenra | mab | humanized | CD125 | Y | asthma |
| Berlimatoxumab[6] | mab | human | Staphylococcus aureus bi-component leukocidin | ? | ||
| Bermekimab | Xilonix | mab | human | IL1A | colorectal cancer | |
| Bersanlimab[4] | mab | human | ICAM-1 | ? | ||
| Bertilimumab[14] | mab | human | CCL11 (eotaxin-1) | severe allergic disorders | ||
| Besilesomab[20] | Scintimun | mab | mouse | CEA-related antigen | inflammatory lesions and metastases (detection) | |
| Bevacizumab[10] | Avastin | mab | humanized | VEGF-A | Y | metastatic cancer, retinopathy of prematurity |
| Bezlotoxumab | Zinplava | mab | human | Clostridium difficile | Y | Clostridium difficile colitis |
| Biciromab | FibriScint | Fab' | mouse | fibrin II, beta chain | thromboembolism (diagnosis) | |
| Bimagrumab[21] | mab | human | ACVR2B | myostatin inhibitor | ||
| Bimekizumab[22] | mab | humanized | IL 17A and IL 17F | ankylosing spondylitis, psoriasis | ||
| Birtamimab | mab | chimeric | serum amyloid A protein | amyloidosis | ||
| Bivatuzumab mertansine | mab | humanized | CD44 v6 | squamous cell carcinoma | ||
| Bleselumab[7] | mab | human | CD40 | organ transplant rejection | ||
| Blinatumomab | Blincyto | BiTE | mouse | CD19 | Y | pre-B ALL (CD19+) |
| Blontuvetmab[23] | Blontress | mab | veterinary | CD20 | ||
| Blosozumab[24] | mab | humanized | SOST | osteoporosis | ||
| Bococizumab | mab | humanized | neural apoptosis-regulated proteinase 1 | dyslipidemia | ||
| Brazikumab[25] | mab | human | IL23 | Crohn's disease | ||
| Brentuximab vedotin | Adcentris | mab | chimeric | CD30 (TNFRSF8) | Y | Hodgkin lymphoma |
| Briakinumab | mab | human | IL-12, IL-23 | psoriasis, rheumatoid arthritis, inflammatory bowel diseases, multiple sclerosis | ||
| Brodalumab | Siliz | mab | human | IL-17 | Y | Plaque psoriasis |
| Brolucizumab[15] | scFv | humanized | VEGFA | wet age-related macular degeneration | ||
| Brontictuzumab[19] | mab | humanized | Notch 1 | cancer | ||
| Burosumab[25] | Crysvita | mab | human | FGF 23 | Y | X-linked hypophosphatemia |
| Cabiralizumab[23] | mab | humanized | CSF1R | metastatic pancreatic cancer | ||
| Camidanlumab tesirine[6] | mab | human | CD25 | B-cell Hodgkin’s lymphoma, non-Hodgkin lymphoma, acute lymphoblastic leukemia, acute myeloid leukemia | ||
| Camrelizumab[13] | mab | humanized | PD-1 | hepatocellular carcinoma | ||
| Canakinumab[26] | Ilaris | mab | human | IL-1 | Y | Cryopyrin-associated periodic syndrome |
| Cantuzumab mertansine | mab | humanized | mucin CanAg | colorectal cancer etc. | ||
| Cantuzumab ravtansine[24] | mab | humanized | MUC1 | cancers | ||
| Caplacizumab[27] | Cablivi | sdAb | humanized | VWF | Y | thrombotic thrombocytopenic purpura, thrombosis |
| Capromab pendetide | Prostascint | mab | mouse | prostatic carcinoma cells | Y | prostate cancer (detection) |
| Carlumab | mab | human | MCP-1 | oncology/immune indications | ||
| Carotuximab[23] | mab | chimeric | endoglin | angiosarcoma | ||
| Catumaxomab[18] | Removab | 3funct | rat/mouse hybrid | EpCAM, CD3 | Y | ovarian cancer, malignant ascites, gastric cancer |
| cBR96-doxorubicin immunoconjugate | mab | humanized | Lewis-Y antigen | cancer | ||
| Cedelizumab | mab | humanized | CD4 | prevention of organ transplant rejections, treatment of autoimmune diseases | ||
| Cemiplimab | Libtayo | mab | human | PCDC1 | Y | cutaneous squamous cell carcinoma |
| Cergutuzumab amunaleukin[7] | mab | humanized | IL2 | cancer | ||
| Certolizumab pegol[5] | Cimzia | Fab' | humanized | TNF-α | Y | Crohn's disease, Rheumatoid arthritis, axial spondyloarthritis, psoriasis arthritis |
| Cetrelimab[4] | mab | human | PD-1 | cancer | ||
| Cetuximab | Erbitux | mab | chimeric | EGFR | Y | metastatic colorectal cancer and head and neck cancer |
| Cibisatamab[4] | mab | humanized | CEACAM5 | cancer | ||
| Cirmtuzumab | ? | humanized | ROR1 | chronic lymphocytic leukemia | ||
| Citatuzumab bogatox[8] | Fab | humanized | EpCAM | ovarian cancer and other solid tumors | ||
| Cixutumumab | mab | human | IGF-1 receptor (CD221) | Y | solid tumors | |
| Clazakizumab[28] | mab | humanized | IL6 | rheumatoid arthritis | ||
| Clenoliximab | mab | chimeric | CD4 | rheumatoid arthritis | ||
| Clivatuzumab tetraxetan | hPAM4-Cide | mab | humanized | MUC1 | pancreatic cancer | |
| Codrituzumab[3] | mab | humanized | glypican 3 | cancer | ||
| Cofetuzumab pelidotin[6] | mab | humanized | PTK7 | cancer | ||
| Coltuximab ravtansine[3] | mab | chimeric | CD19 | cancer | ||
| Conatumumab[8] | mab | human | TRAIL-R2 | cancer | ||
| Concizumab[21] | mab | humanized | TFPI | bleeding | ||
| Cosfroviximab[17] | ZMapp | mab | chimeric | ebolavirus glycoprotein | Ebola virus | |
| Crenezumab | mab | humanized | 1-40-β-amyloid | Alzheimer's disease | ||
| Crizanlizumab[13] | mab | humanized | selectin P | sickle-cell disease | ||
| Crotedumab[23] | mab | human | GCGR | diabetes | ||
| CR6261 | mab | human | Influenza A hemagglutinin | infectious disease/influenza A | ||
| Cusatuzumab[4] | mab | humanized | CD70 | cancer | ||
| Dacetuzumab[9] | mab | humanized | CD40 | hematologic cancers | ||
| Daclizumab | Zenapax | mab | humanized | CD25 (α chain of IL-2 receptor) | Y | prevention of organ transplant rejections, multiple sclerosis |
| Dalotuzumab | mab | humanized | IGF-1 receptor (CD221) | cancer etc. | ||
| Dapirolizumab pegol[22] | mab | humanized | CD154 (CD40L) | ? | ||
| Daratumumab[29] | Darzalex | mab | human | CD38 | Y | Multiple myeloma |
| Dectrekumab[15] | mab | human | IL-13 | ? | ||
| Demcizumab | mab | humanized | DLL4 | cancer | ||
| Denintuzumab mafodotin[19] | mab | humanized | CD19 | cancer | ||
| Denosumab[30] | Prolia | mab | human | RANKL | Y | osteoporosis, bone metastases etc. |
| Depatuxizumab mafodotin[25] | mab | chimeric/humanized | EGFR | glioblastoma | ||
| Derlotuximab biotin | mab | chimeric | histone complex | recurrent glioblastoma multiforme | ||
| Detumomab | mab | mouse | B-lymphoma cell | lymphoma | ||
| Dezamizumab[13] | mab | humanized | serum amyloid P component | ? | ||
| Dinutuximab | Unituxin | mab | chimeric | GD2 ganglioside | Y | neuroblastoma |
| Diridavumab | mab | human | hemagglutinin | influenza A | ||
| Domagrozumab[23] | mab | humanized | GDF-8 | Duchenne muscular dystrophy | ||
| Dorlimomab aritox[31] | F(ab')2 | mouse | ? | ? | ||
| Dostarlimab[32] | mab | humanized | PCDP1 | cancer | ||
| Drozitumab | mab | human | DR5 | cancer etc. | ||
| DS-8201 | ? | humanized | HER2 | gastric or gastroesophageal junction adenocarcinoma | ||
| Duligotuzumab[33] | mab | humanized | ERBB3 (HER3) | testicular cancer | ||
| Dupilumab | Dupixent | mab | human | IL4 | Y | atopic diseases |
| Durvalumab | Imfinzi | mab | human | PD-L1 | Y | cancer |
| Dusigitumab | mab | human | ILGF2 | B-cell malignancies | ||
| Duvortuxizumab[17] | scFv | chimeric/humanized | CD19, CD3E | cancer | ||
| Ecromeximab[12] | mab | chimeric | GD3 ganglioside | malignant melanoma | ||
| Eculizumab[12] | Soliris | mab | humanized | C5 | Y | paroxysmal nocturnal hemoglobinuria, atypical HUS |
| Edobacomab | mab | mouse | endotoxin | sepsis caused by Gram-negative bacteria | ||
| Edrecolomab | Panorex | mab | mouse | EpCAM | colorectal carcinoma | |
| Efalizumab[34] | Raptiva | mab | humanized | LFA-1 (CD11a) | psoriasis (blocks T-cell migration) | |
| Efungumab[2] | Mycograb | scFv | human | Hsp90 | invasive Candida infection | |
| Eldelumab | mab | human | interferon gamma-induced protein | Crohn's disease, ulcerative colitis | ||
| Elezanumab[13] | mab | human | RGMA | spinal cord injury and multiple sclerosis | ||
| Elgemtumab[15] | mab | human | ERBB3 (HER3) | cancer | ||
| Elotuzumab | Empliciti | mab | humanized | SLAMF7 | Y | multiple myeloma |
| Elsilimomab | mab | mouse | IL-6 | ? | ||
| Emactuzumab[19] | mab | humanized | CSF1R | cancer | ||
| Emapalumab | Gamifant | mab | human | interferon gamma | hemophagocytic lymphohistiocytosis | |
| Emibetuzumab | mab | humanized | HHGFR | cancer | ||
| Emicizumab[7] | Hemlibra | mab | humanized | activated F9, F10 | Y | haemophilia A |
| Enapotamab vedotin[4] | mab | human | AXL | cancer | ||
| Enavatuzumab | mab | humanized | TWEAK receptor | cancer etc. | ||
| Enfortumab vedotin | mab | human | nectin-4 | urothelial cancer | ||
| Enlimomab pegol[35] | mab | mouse | ICAM-1 (CD54) | ? | ||
| Enoblituzumab | mab | humanized | CD276 | cancer | ||
| Enokizumab | mab | humanized | IL9 | asthma | ||
| Enoticumab[33] | mab | human | DLL4 | ? | ||
| Ensituximab | mab | chimeric | 5AC | cancer | ||
| Epitumomab cituxetan[36] | mab | mouse | episialin | ? | ||
| Epratuzumab | mab | humanized | CD22 | cancer, SLE | ||
| Eptinezumab | mab | humanized | calcitonin gene-related peptide | migraine | ||
| Erenumab[25] | Aimovig | mab | human | CGRP | Y | migraine |
| Erlizumab[37] | F(ab')2 | humanized | ITGB2 (CD18) | heart attack, stroke, traumatic shock | ||
| Ertumaxomab[18] | Rexomun | 3funct | rat/mouse hybrid | HER2/neu, CD3 | Y | breast cancer etc. |
| Etaracizumab | Abegrin | mab | humanized | integrin αvβ3 | Y | melanoma, prostate cancer, ovarian cancer etc. |
| Etigilimab[4] | mab | humanized | TIGIT | ? | ||
| Etrolizumab | mab | humanized | integrin β7 | inflammatory bowel disease | ||
| Evinacumab | mab | human | angiopoietin 3 | dyslipidemia | ||
| Evolocumab[21] | Repatha | mab | human | PCSK9 | Y | hypercholesterolemia |
| Exbivirumab[38] | mab | human | hepatitis B surface antigen | hepatitis B | ||
| Fanolesomab[39] | NeutroSpec | mab | mouse | CD15 | appendicitis (diagnosis) | |
| Faralimomab | mab | mouse | interferon receptor | ? | ||
| Faricimab[4] | mab | humanized | VEGF-A and Ang-2 | angiogenesis, ocular vascular diseases | ||
| Farletuzumab | mab | humanized | folate receptor 1 | ovarian cancer | ||
| Fasinumab | mab | human | HNGF | acute sciatic pain | ||
| FBTA05[40][41] | Lymphomun | 3funct | rat/mouse hybrid | CD20 | chronic lymphocytic leukaemia | |
| Felvizumab | mab | humanized | respiratory syncytial virus | respiratory syncytial virus infection | ||
| Fezakinumab[42][43] | mab | human | IL-22 | rheumatoid arthritis, psoriasis | ||
| Fibatuzumab[7] | mab | humanized | ephrin receptor A3 | ? | ||
| Ficlatuzumab | mab | humanized | HGF | cancer etc. | ||
| Figitumumab | mab | human | IGF-1 receptor (CD221) | adrenocortical carcinoma, non-small cell lung carcinoma etc. | ||
| Firivumab[19] | mab | human | influenza A virus hemagglutinin | ? | ||
| Flanvotumab | mab | human | TYRP1 (glycoprotein 75) | melanoma | ||
| Fletikumab | mab | human | IL 20 | rheumatoid arthritis | ||
| Flotetuzumab[4] | di-scFv | humanized | IL 3 receptor | hematological malignancies | ||
| Fontolizumab[12] | HuZAF | mab | humanized | IFN-γ | Crohn's disease etc. | |
| Foralumab[44] | mab | human | CD3 epsilon | ? | ||
| Foravirumab[8] | mab | human | rabies virus glycoprotein | rabies (prophylaxis) | ||
| Fremanezumab[13] | Ajovy | mab | humanized | calcitonin gene-related peptide alpha | Y | migraine and cluster headache |
| Fresolimumab | mab | human | TGF-β | idiopathic pulmonary fibrosis, focal segmental glomerulosclerosis, cancer | ||
| Frovocimab[32] | mab | humanized | PCSK9 | hypercholesterolemia | ||
| Frunevetmab[17] | mab | veterinary | NGF | ? | ||
| Fulranumab | mab | human | NGF | pain | ||
| Futuximab[33] | mab | chimeric | EGFR | cancer | ||
| Galcanezumab[23] | Emgality | mab | humanized | calcitonin | Y | migraine |
| Galiximab | mab | chimeric | CD80 | B-cell lymphoma | ||
| Gancotamab | scFv | human | HER2/neu | cancer | ||
| Ganitumab | mab | human | IGF-1 receptor (CD221) | cancer | ||
| Gantenerumab[26] | mab | human | beta amyloid | Alzheimer's disease | ||
| Gatipotuzumab[17] | mab | humanized | MUC1 | cancer | ||
| Gavilimomab[37] | mab | mouse | CD147 (basigin) | graft versus host disease | ||
| Gedivumab[17] | mab | human | hemagglutinin HA | ? | ||
| Gemtuzumab ozogamicin | Mylotarg | mab | humanized | CD33 | Y | acute myelogenous leukemia |
| Gevokizumab | mab | humanized | IL-1β | diabetes etc. | ||
| Gilvetmab[17] | mab | veterinary | PCDC1 | ? | ||
| Gimsilumab[6] | mab | human | CSF2 | rheumatoid arthritis | ||
| Girentuximab[29] | Rencarex | mab | chimeric | carbonic anhydrase 9 (CA-IX) | Y | clear cell renal cell carcinoma[45] |
| Glembatumumab vedotin[46][47] | mab | human | GPNMB | melanoma, breast cancer | ||
| Golimumab[38] | Simponi | mab | human | TNF-α | Y | rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis |
| Gomiliximab | mab | chimeric | CD23 (IgE receptor) | allergic asthma | ||
| Gosuranemab | mab | humanized | tau protein | progressive supranuclear palsy | ||
| Guselkumab | Tremfya | mab | human | IL23 | Y | psoriasis |
| Ianalumab[6] | mab | human | BAFF-R | autoimmune hepatitis | ||
| Ibalizumab[26] | Trogarzo | mab | humanized | CD4 | Y | HIV infection |
| IBI308 | ? | human | PD-1 | squamous cell non-small cell lung cancer | ||
| Ibritumomab tiuxetan | Zevalin | mab | mouse | CD20 | Y | non-Hodgkin's lymphoma |
| Icrucumab | mab | human | VEGFR-1 | cancer etc. | ||
| Idarucizumab[3] | Praxbind | mab | humanized | dabigatran | Y | reversal of anticoagulant effects of dabigatran |
| Ifabotuzumab[13] | mab | humanized | EPHA3 | ? | ||
| Igovomab | Indimacis-125 | F(ab')2 | mouse | CA-125 | ovarian cancer (diagnosis) | |
| Iladatuzumab vedotin[6] | mab | humanized | CD97B | cancer | ||
| IMAB362 | mab | human | CLDN18.2 | gastrointestinal adenocarcinomas and pancreatic tumor | ||
| Imalumab[19] | mab | human | MIF | cancer | ||
| Imaprelimab[4] | mab | humanized | MCAM | ? | ||
| Imciromab | Myoscint | mab | mouse | cardiac myosin | Y | cardiac imaging |
| Imgatuzumab[33] | mab | humanized | EGFR | cancer | ||
| Inclacumab[27] | mab | human | selectin P | cardiovascular disease | ||
| Indatuximab ravtansine[24] | mab | chimeric | SDC1 | cancer | ||
| Indusatumab vedotin[15] | mab | human | GUCY2C | cancer | ||
| Inebilizumab[7] | mab | humanized | CD19 | cancer, systemic sclerosis, multiple sclerosis | ||
| Infliximab | Remicade | mab | chimeric | TNF-α | Y | rheumatoid arthritis, ankylosing spondylitis, psoriatic arthritis, psoriasis, Crohn's disease, ulcerative colitis |
| Intetumumab[48][49] | mab | human | CD51 | solid tumors (prostate cancer, melanoma) | ||
| Inolimomab | mab | mouse | CD25 (α chain of IL-2 receptor) | graft versus host disease | ||
| Inotuzumab ozogamicin[20] | Besponsa | mab | humanized | CD22 | ALL | |
| Ipilimumab[30] | Yervoy | mab | human | CD152 | Y | melanoma |
| Iomab-B | ? | mouse | CD45 | ablation of bone marrow | ||
| Iratumumab[30] | mab | human | CD30 (TNFRSF8) | Hodgkin's lymphoma | ||
| Isatuximab | mab | chimeric | CD38 | multiple myeloma | ||
| Iscalimab[4] | mab | human | CD40 | ? | ||
| Istiratumab[6] | mab | human | IGF1R, CD221 | advanced solid tumors | ||
| Itolizumab[44] | Alzumab | mab | humanized | CD6 | Y | psoriasis |
| Ixekizumab | Taltz | mab | humanized | IL 17A | Y | autoimmune diseases |
| Keliximab | mab | chimeric | CD4 | chronic asthma | ||
| Labetuzumab[34] | CEA-Cide | mab | humanized | CEA | colorectal cancer | |
| Lacnotuzumab[17] | mab | humanized | CSF1, MCSF | cancer | ||
| Ladiratuzumab vedotin[6] | mab | humanized | LIV-1 | cancer | ||
| Lampalizumab[33] | Fab | humanized | CFD | geographic atrophy secondary to age-related macular degeneration | ||
| Lanadelumab[23] | Takhzyro | mab | human | kallikrein | Y | angioedema |
| Landogrozumab[7] | mab | humanized | GDF-8 | muscle wasting disorders | ||
| Laprituximab emtansine[23] | mab | chimeric | EGFR | ? | ||
| Larcaviximab[17] | mab | chimeric | ebolavirus glycoprotein | Ebola virus | ||
| Lebrikizumab | mab | humanized | IL-13 | asthma | ||
| Lemalesomab[37] | mab | mouse | NCA-90 (granulocyte antigen) | diagnostic agent | ||
| Lendalizumab[23] | mab | humanized | C5 | ? | ||
| Lenvervimab[4] | mab | humanized | hepatitis B surfage antigen | hepatitis B | ||
| Lenzilumab[19] | mab | human | CSF2 | chronic myelomonocytic leukemia and juvenile myelomonocytic leukemia | ||
| Lerdelimumab[10] | mab | human | TGF beta 2 | reduction of scarring after glaucoma surgery | ||
| Leronlimab[4] | mab | humanized | CCR5 | ? | ||
| Lesofavumab[17] | mab | human | hemagglutinin HA | ? | ||
| Letolizumab[17] | scFv | humanized | TRAP | inflammatory diseases | ||
| Lexatumumab[2] | mab | human | TRAIL-R2 | cancer | ||
| Libivirumab[38] | mab | human | hepatitis B surface antigen | hepatitis B | ||
| Lifastuzumab vedotin | mab | humanized | phosphate-sodium co-transporter | cancer | ||
| Ligelizumab[33] | mab | humanized | IGHE | severe asthma and chronic spontaneous urticaria | ||
| Loncastuximab tesirine[6] | mab | chimeric | CD19 | cancer | ||
| Losatuxizumab vedotin[17] | mab | chimeric/humanized | EGRF, ERBB1 HER1 | cancer | ||
| Lilotomab satetraxetan[15] | mab | mouse | CD37 | cancer | ||
| Lintuzumab | mab | humanized | CD33 | cancer | ||
| Lirilumab[33] | mab | human | KIR2D | solid and hematological cancers | ||
| Lodelcizumab[21] | mab | humanized | PCSK9 | hypercholesterolemia | ||
| Lokivetmab[15] | Cytopoint[50] | mab | veterinary | Canis lupus familiaris IL31 | Y | clinical signs of atopic dermatitis in dogs[50] |
| Lorvotuzumab mertansine | mab | humanized | CD56 | cancer | ||
| Lucatumumab[9] | mab | human | CD40 | multiple myeloma, non-Hodgkin's lymphoma, Hodgkin's lymphoma | ||
| Lulizumab pegol | mab | humanized | CD28 | autoimmune diseases | ||
| Lumiliximab[5] | mab | chimeric | CD23 (IgE receptor) | chronic lymphocytic leukemia | ||
| Lumretuzumab[19] | mab | humanized | ERBB3 (HER3) | cancer | ||
| Lupartumab amadotin[13] | mab | human | LYPD3 | ? | ||
| Lutikizumab[13] | mab | humanized | interleukin 1 alpha | ? | ||
| Mapatumumab[18] | mab | human | TRAIL-R1 | cancer | ||
| Margetuximab | mab | humanized | HER2 | breast cancer | ||
| Marstacimab | mab | human | TFPI | bleeding with hemophilia | ||
| Maslimomab | ? | mouse | T-cell receptor | |||
| Mavrilimumab | mab | human | GMCSF receptor α-chain | rheumatoid arthritis | ||
| Matuzumab[14] | mab | humanized | EGFR | colorectal, lung and stomach cancer | ||
| Mepolizumab | Bosatria | mab | humanized | IL-5 | Y | asthma and white blood cell diseases |
| Metelimumab[39] | mab | human | TGF beta 1 | systemic scleroderma | ||
| Milatuzumab[9] | mab | humanized | CD74 | multiple myeloma and other hematological malignancies | ||
| Minretumomab | mab | mouse | TAG-72 | tumor detection (and therapy?) | ||
| Mirikizumab[6] | mab | humanized | IL23A | psoriasis | ||
| Mirvetuximab soravtansine | mab | chimeric | folate receptor alpha | ovarian cancer | ||
| Mitumomab | mab | mouse | GD3 ganglioside | small cell lung carcinoma | ||
| Modotuximab | mab | chimeric | EGFR extracellular domain III | cancer | ||
| Mogamulizumab | Poteligeo | mab | humanized | CCR4 | Y | adult T-cell leukemia/lymphoma |
| Monalizumab[7] | mab | humanized | NKG2A | rheumatoid arthritis, gynecologic malignancies, and other cancers | ||
| Morolimumab | mab | human | Rhesus factor | |||
| Mosunetuzumab[6] | mab | humanized | CD3E, MS4A1, CD20 | cancer | ||
| Motavizumab[2] | Numax | mab | humanized | respiratory syncytial virus | respiratory syncytial virus (prevention) | |
| Moxetumomab pasudotox | Lumoxiti | mab | mouse | CD22 | Y | hairy cell leukemia |
| Muromonab-CD3 | Orthoclone OKT3 | mab | mouse | CD3 | prevention of organ transplant rejections | |
| Nacolomab tafenatox | Fab | mouse | C242 antigen | colorectal cancer | ||
| Namilumab[16] | mab | human | CSF2 | |||
| Naptumomab estafenatox[51] | Fab | mouse | 5T4 | non-small cell lung carcinoma, renal cell carcinoma | ||
| Naratuximab emtansine[23] | mab | chimeric | CD37 | |||
| Narnatumab | mab | human | RON | cancer | ||
| Natalizumab | Tysabri | mab | humanized | integrin α4 | Y | multiple sclerosis, Crohn's disease |
| Navicixizumab[23] | mab | chimeric/humanized | DLL4 | cancer | ||
| Navivumab[7] | mab | human | influenza A virus hemagglutinin HA | |||
| Naxitamab | ? | humanized | c-Met | high-risk neuroblastoma and refractory osteomedullary disease | ||
| Nebacumab | mab | human | endotoxin | sepsis | ||
| Necitumumab[52] | Portrazza | mab | human | EGFR | Y | non-small cell lung carcinoma |
| Nemolizumab[15] | mab | humanized | IL31RA | eczema[53] | ||
| NEOD001 | ? | humanized | amyloid | primary systemic amyloidosis | ||
| Nerelimomab | mab | mouse | TNF-α | ? | ||
| Nesvacumab | mab | human | angiopoietin 2 | cancer | ||
| Netakimab[4] | mab | chimeric | interleukin 17A | ? | ||
| Nimotuzumab[30][54] | Theracim, Theraloc | mab | humanized | EGFR | Y | squamous cell carcinoma, head and neck cancer, nasopharyngeal cancer, glioma |
| Nirsevimab | mab | human | RSVFR | respiratory syncytial virus | ||
| Nivolumab | Opdivo | mab | human | PD-1 | Y | cancer |
| Nofetumomab merpentan | Verluma | Fab | mouse | ? | cancer (diagnosis) | |
| Obiltoxaximab | Anthim | mab | chimeric | Bacillus anthracis anthrax | Y | Bacillus anthracis spores |
| Obinutuzumab | Gazyva | mab | humanized | CD20 | Y | Chronic lymphatic leukemia |
| Ocaratuzumab | mab | humanized | CD20 | cancer | ||
| Ocrelizumab[30] | Ocrevus | mab | humanized | CD20 | Y | rheumatoid arthritis, lupus erythematosus etc. |
| Odulimomab | mab | mouse | LFA-1 (CD11a) | prevention of organ transplant rejections, immunological diseases | ||
| Ofatumumab[18] | Arzerra | mab | human | CD20 | Y | chronic lymphocytic leukemia etc. |
| Olaratumab | Lartruvo | mab | human | PDGF-R α | Y | cancer |
| Oleclumab[17] | mab | human | 5'-nucleotidase | pancreatic and colorectal cancer | ||
| Olendalizumab | mab | humanized | complement C5a | systemic lupus erythematosus, lupus nephritis, acute graft-versus-hose disease | ||
| Olokizumab[44] | mab | humanized | IL6 | rheumatoid arthritis | ||
| Omalizumab[37] | Xolair | mab | humanized | IgE Fc region | Y | allergic asthma |
| Omburtamab[32] | mab | mouse | CD276 | cancer | ||
| OMS721 | ? | human | MASP-2 | atypical hemolytic uremic syndrome | ||
| Onartuzumab | mab | humanized | human scatter factor receptor kinase | cancer | ||
| Ontuxizumab | mab | chimeric/humanized | TEM1 | cancer | ||
| Onvatilimab[4] | mab | human | VSIR | ? | ||
| Opicinumab | mab | human | LINGO-1 | multiple sclerosis | ||
| Oportuzumab monatox[52] | Vicinium | scFv | humanized | EpCAM | bladder cancer | |
| Oregovomab[39] | OvaRex | mab | mouse | CA-125 | ovarian cancer | |
| Orticumab[33] | mab | human | oxLDL | |||
| Otelixizumab[9] | mab | chimeric/humanized | CD3 | diabetes mellitus type 1 | ||
| Otilimab | mab | human | GMCSF | osteoarthritis, rheumatoid arthritis | ||
| Otlertuzumab | mab | humanized | CD37 | cancer | ||
| Oxelumab | mab | human | OX-40 | asthma | ||
| Ozanezumab | mab | humanized | NOGO-A | ALS and multiple sclerosis | ||
| Ozoralizumab | mab | humanized | TNF-α | inflammation | ||
| Pagibaximab[18] | mab | chimeric | lipoteichoic acid | sepsis (Staphylococcus) | ||
| Palivizumab | Synagis, Abbosynagis | mab | humanized | F protein of respiratory syncytial virus | Y | respiratory syncytial virus (prevention) |
| Pamrevlumab[7] | mab | human | CTGF | idiopathic pulmonary fibrosis (IPF), pancreatic cancer | ||
| Panitumumab[38] | Vectibix | mab | human | EGFR | Y | colorectal cancer |
| Pankomab | mab | humanized | tumor specific glycosylation of MUC1 | ovarian cancer | ||
| Panobacumab[52] | mab | human | Pseudomonas aeruginosa | Pseudomonas aeruginosa infection | ||
| Parsatuzumab[33] | mab | human | EGFL7 | cancer | ||
| Pascolizumab[12] | mab | humanized | IL-4 | asthma | ||
| Pasotuxizumab[19] | mab | chimeric/humanized | folate hydrolase | cancer | ||
| Pateclizumab[24] | mab | humanized | LTA | TNF | ||
| Patritumab[27] | mab | human | ERBB3 (HER3) | cancer | ||
| PDR001 | ? | humanized | PD-1 | melanoma | ||
| Pembrolizumab | Keytruda | mab | humanized | PD-1 | Y | melanoma and other cancers |
| Pemtumomab | Theragyn | ? | mouse | MUC1 | cancer | |
| Perakizumab[33] | mab | humanized | IL 17A | arthritis | ||
| Pertuzumab | Omnitarg | mab | humanized | HER2/neu | Y | cancer |
| Pexelizumab[34] | scFv | humanized | C5 | reduction of side effects of cardiac surgery | ||
| Pidilizumab | mab | humanized | PD-1 | cancer and infectious diseases | ||
| Pinatuzumab vedotin[21] | mab | humanized | CD22 | cancer | ||
| Pintumomab | mab | mouse | adenocarcinoma antigen | adenocarcinoma (imaging) | ||
| Placulumab | mab | human | human TNF | pain and inflammatory diseases | ||
| Plozalizumab[7] | mab | humanized | CCR2 | diabetic nephropathy and arteriovenous graft patency | ||
| Pogalizumab[23] | mab | humanized | TNFR superfamily member 4 | ? | ||
| Polatuzumab vedotin[21] | mab | humanized | CD79B | diffuse large B-cell lymphoma | ||
| Ponezumab | mab | humanized | human beta-amyloid | Alzheimer's disease | ||
| Porgaviximab[17] | mab | chimeric | Zaire ebolavirus glycoprotein | Ebola virus disease | ||
| Prasinezumab[6] | mab | humanized | NACP | Parkinson's disease | ||
| Prezalizumab[23] | mab | humanized | ICOSL | ? | ||
| Priliximab | mab | chimeric | CD4 | Crohn's disease, multiple sclerosis | ||
| Pritoxaximab[21] | mab | chimeric | E. coli shiga toxin type-1 | ? | ||
| Pritumumab | mab | human | vimentin | brain cancer | ||
| PRO 140 | ? | humanized | CCR5 | HIV infection | ||
| Quilizumab[27] | mab | humanized | IGHE | asthma | ||
| Racotumomab[52] | Vaxira | mab | mouse | NGNA ganglioside | Y | non-small cell lung cancer |
| Radretumab[16] | mab | human | fibronectin extra domain-B | cancer | ||
| Rafivirumab[8] | mab | human | rabies virus glycoprotein | rabies (prophylaxis) | ||
| Ralpancizumab | mab | humanized | neural apoptosis-regulated proteinase 1 | dyslipidemia | ||
| Ramucirumab | Cyramza | mab | human | VEGFR2 | Y | solid tumors |
| Ranevetmab | mab | veterinary | NGF | osteoarthritis in dogs | ||
| Ranibizumab[5] | Lucentis | Fab | humanized | VEGF-A | Y | macular degeneration (wet form) |
| Raxibacumab[20] | mab | human | anthrax toxin, protective antigen | Y | anthrax (prophylaxis and treatment) | |
| Ravagalimab[4] | mab | humanized | CD40 | ? | ||
| Ravulizumab[6] | mab | humanized | C5 | paroxysmal nocturnal hemaglobinuria, atypical hemolytic uremic syndrome | ||
| Refanezumab | mab | humanized | myelin-associated glycoprotein | recovery of motor function after stroke | ||
| Regavirumab | mab | human | cytomegalovirus glycoprotein B | cytomegalovirus infection | ||
| Relatlimab | mab | human | LAG3 | melanoma | ||
| Remtolumab[13] | mab | human | interleukin 17 alpha, TNF | ? | ||
| Reslizumab[34] | Cinqair | mab | humanized | IL-5 | Y | inflammations of the airways, skin and gastrointestinal tract |
| Rilotumumab | mab | human | HGF | solid tumors | ||
| Rinucumab | mab | human | platelet-derived growth factor receptor beta | neovascular age-related macular degeneration | ||
| Risankizumab[7] | mab | humanized | IL23A | Crohn's disease, psoriasis, psoriatic arthritis, and asthma | ||
| Rituximab | MabThera, Rituxan | mab | chimeric | CD20 | Y | lymphomas, leukemias, some autoimmune disorders |
| Rivabazumab pegol[7] | mab | humanized | Pseudomonas aeruginosa type III secretion system | ? | ||
| Robatumumab | mab | human | IGF-1 receptor (CD221) | cancer | ||
| Rmab | RabiShield | ? | human | rabies virus G glycoprotein | Y | post-exposure prophylaxis of rabies |
| Roledumab[44] | mab | human | RHD | Rh disease | ||
| Romilkimab[4] | mab | chimeric/humanized | interleukin 13 | ? | ||
| Romosozumab | Evenity | mab | humanized | sclerostin | osteoporosis | |
| Rontalizumab | mab | humanized | IFN-α | systemic lupus erythematosus | ||
| Rosmantuzumab | mab | humanized | root plate-specific spondin 3 | cancer | ||
| Rovalpituzumab tesirine[7] | mab | humanized | DLL3 | small cell lung cancer | ||
| Rovelizumab | LeukArrest | mab | humanized | CD11, CD18 | Y | haemorrhagic shock etc. |
| Rozanolixizumab[13] | mab | chimeric/humanized | FCGRT | ? | ||
| Ruplizumab[10] | Antova | mab | humanized | CD154 (CD40L) | Y | rheumatic diseases |
| SA237 | ? | humanized | IL-6R | neuromyelitis optica and neuromyelitis optica spectrum disorders | ||
| Sacituzumab govitecan | mab | humanized | TROP-2 | triple-negative breast cancer | ||
| Samalizumab | mab | humanized | CD200 | cancer | ||
| Samrotamab vedotin[4] | mab | chimeric/humanized | LRRC15 | cancer | ||
| Sarilumab | Kevzara | mab | human | IL6 | Y | rheumatoid arthritis, ankylosing spondylitis |
| Satralizumab | mab | humanized | IL6 receptor | neuromyelitis optica | ||
| Satumomab pendetide | mab | mouse | TAG-72 | cancer (diagnosis) | ||
| Secukinumab | Cosentyx | mab | human | IL 17A | Y | uveitis, rheumatoid arthritis psoriasis |
| Selicrelumab[17] | mab | human | CD40 | ? | ||
| Seribantumab[21] | mab | human | ERBB3 (HER3) | cancer | ||
| Setoxaximab[21] | mab | chimeric | E. coli shiga toxin type-2 | ? | ||
| Setrusumab[6] | mab | human | SOST | ? | ||
| Sevirumab | ? | human | cytomegalovirus | cytomegalovirus infection | ||
| Sibrotuzumab | mab | humanized | FAP | cancer | ||
| SGN-CD19A | mab | humanized | CD19 | acute lymphoblastic leukemia and B-cell non-Hodgkin lymphoma | ||
| SHP647 | ? | human | mucosal addressin cell adhesion molecule | Crohn's disease | ||
| Sifalimumab | mab | humanized | IFN-α | SLE, dermatomyositis, polymyositis | ||
| Siltuximab | Sylvant | mab | chimeric | IL-6 | Y | cancer |
| Simtuzumab[33] | mab | humanized | LOXL2 | fibrosis | ||
| Siplizumab[12] | mab | humanized | CD2 | psoriasis, graft-versus-host disease (prevention) | ||
| Sirtratumab vedotin[6] | mab | human | SLITRK6 | cancer | ||
| Sirukumab | mab | human | IL-6 | rheumatoid arthritis | ||
| Sofituzumab vedotin | mab | humanized | CA-125 | ovarian cancer | ||
| Solanezumab[52] | mab | humanized | beta amyloid | Alzheimer's disease | ||
| Solitomab[27] | BiTE | mouse | EpCAM | gastrointestinal, lung, and other cancers | ||
| Sonepcizumab[55] | ? | humanized | sphingosine-1-phosphate | choroidal and retinal neovascularization | ||
| Sontuzumab[54] | mab | humanized | episialin | |||
| Spartalizumab[6] | mab | humanized | PDCD1, CD279 | melanoma | ||
| Stamulumab[30][54] | mab | human | myostatin | muscular dystrophy | ||
| Sulesomab | LeukoScan | Fab' | mouse | NCA-90 (granulocyte antigen) | osteomyelitis (imaging) | |
| Suptavumab | mab | human | RSVFR | medically attended lower respiratory disease | ||
| Sutimlimab[4] | mab | chimeric/humanized | C1s | cold agglutinin disease | ||
| Suvizumab[46] | mab | humanized | HIV-1 | viral infections | ||
| Suvratoxumab[17] | mab | human | Staphylococcus aureus alpha toxin | nosocomial pneumonia | ||
| Tabalumab | mab | human | BAFF | B-cell cancers | ||
| Tacatuzumab tetraxetan | AFP-Cide | mab | humanized | alpha-fetoprotein | cancer | |
| Tadocizumab[54] | Fab | humanized | integrin αIIbβ3 | percutaneous coronary intervention | ||
| Talacotuzumab[6] | mab | humanized | CD123 | ? | ||
| Talizumab | mab | humanized | IgE | allergic reaction | ||
| Tamtuvetmab[23] | Tactress | mab | veterinary | CD52 | ||
| Tanezumab[8] | mab | humanized | NGF | pain | ||
| Taplitumomab paptox[37] | mab | mouse | CD19 | cancer | ||
| Tarextumab | mab | human | Notch receptor | cancer | ||
| Tavolimab | mab | chimeric/humanized | CD134 | cancer | ||
| Tefibazumab[20] | Aurexis | mab | humanized | clumping factor A | Staphylococcus aureus infection | |
| Telimomab aritox | Fab | mouse | ? | |||
| Telisotuzumab vedotin | mab | humanized | HGFR | cancer | ||
| Tenatumomab[9] | mab | mouse | tenascin C | cancer | ||
| Teneliximab[12] | mab | chimeric | CD40 | autoimmune diseases and prevention of organ transplant rejection | ||
| Teplizumab[26] | mab | humanized | CD3 | diabetes mellitus type 1 | ||
| Tepoditamab[4] | mab | human | dendritic cell-associated lectin 2 | cancer | ||
| Teprotumumab | mab | human | IGF-1 receptor (CD221) | thyroid eye disease | ||
| Tesidolumab[15] | mab | human | C5 | |||
| Tetulomab | mab | humanized | CD37 | cancer[56] | ||
| Tezepelumab[7] | mab | human | TSLP | asthma, atopic dermatitis | ||
| TGN1412 | ? | humanized | CD28 | chronic lymphocytic leukemia, rheumatoid arthritis | ||
| Tibulizumab[6] | mab | humanized | BAFF | autoimmune disorders | ||
| Tildrakizumab | Ilumya | mab | humanized | IL23 | Y | immunologically mediated inflammatory disorders |
| Tigatuzumab[9] | mab | humanized | TRAIL-R2 | cancer | ||
| Timigutuzumab[17] | mab | humanized | HER2 | cancer | ||
| Timolumab[23] | mab | human | AOC3 | |||
| Tiragotumab[6] | mab | human | TIGIT | cancer | ||
| Tislelizumab[6] | mab | humanized | PCDC1, CD279 | non-small cell lung cancer | ||
| Tisotumab vedotin[7] | mab | human | coagulation factor III | relapsed or refractory cervical cancer[57] | ||
| TNX-650 | ? | humanized | IL-13 | Hodgkin's lymphoma | ||
| Tocilizumab[5] | Actemra, RoActemra | mab | humanized | IL-6 receptor | Y | rheumatoid arthritis |
| Tomuzotuximab[17] | mab | humanized | EGFR, HER1 | cancer | ||
| Toralizumab[12] | mab | humanized | CD154 (CD40L) | rheumatoid arthritis, lupus nephritis etc. | ||
| Tosatoxumab[3] | mab | human | Staphylococcus aureus | |||
| Tositumomab | Bexxar | ? | mouse | CD20 | Y | follicular lymphoma |
| Tovetumab | mab | human | CD140a | cancer | ||
| Tralokinumab | mab | human | IL-13 | asthma, atopic dermatitis | ||
| Trastuzumab | Herceptin | mab | humanized | HER2/neu | Y | breast cancer |
| Trastuzumab emtansine | Kadcyla | mab | humanized | HER2/neu | Y | breast cancer |
| TRBS07[58] | Ektomab | 3funct | ? | GD2 ganglioside | melanoma | |
| Tregalizumab[16] | mab | humanized | CD4 | |||
| Tremelimumab | mab | human | CTLA-4 | non-small cell lung, head & neck, urothelial cancer | ||
| Trevogrumab | mab | human | growth differentiation factor 8 | muscle atrophy due to orthopedic disuse and sarcopenia | ||
| Tucotuzumab celmoleukin[30][54] | mab | humanized | EpCAM | cancer | ||
| Tuvirumab | ? | human | hepatitis B virus | chronic hepatitis B | ||
| Ublituximab[16] | mab | chimeric | MS4A1 | multiple sclerosis, chronic lymphocytic leukemia | ||
| Ulocuplumab | mab | human | CXCR4 (CD184) | hematologic malignancies | ||
| Urelumab | mab | human | 4-1BB (CD137) | cancer etc. | ||
| Urtoxazumab[5] | mab | humanized | Escherichia coli | diarrhoea caused by E. coli | ||
| Ustekinumab[8] | Stelara | mab | human | IL-12, IL-23 | Y | multiple sclerosis, psoriasis, psoriatic arthritis |
| Utomilumab[25] | mab | human | 4-1BB (CD137) | diffuse large B-cell lymphoma | ||
| Vadastuximab talirine[7] | mab | chimeric | CD33 | Acute myeloid leukemia | ||
| Vanalimab[4] | mab | humanized | CD40 | ? | ||
| Vandortuzumab vedotin[15] | mab | humanized | STEAP1 | cancer | ||
| Vantictumab | mab | human | Frizzled receptor | cancer | ||
| Vanucizumab[19] | mab | humanized | angiopoietin 2 | cancer | ||
| Vapaliximab[12] | mab | chimeric | AOC3 (VAP-1) | |||
| Varisacumab[17] | mab | human | VEGF-A | angiogenesis | ||
| Varlilumab | mab | human | CD27 | solid tumors and hematologic malignancies | ||
| Vatelizumab[24] | mab | humanized | ITGA2 (CD49b) | |||
| Vedolizumab | Entyvio | mab | humanized | integrin α4β7 | Y | Crohn's disease, ulcerative colitis |
| Veltuzumab[9] | mab | humanized | CD20 | non-Hodgkin's lymphoma | ||
| Vepalimomab | mab | mouse | AOC3 (VAP-1) | inflammation | ||
| Vesencumab[16] | mab | human | NRP1 | solid malignancies | ||
| Visilizumab[37] | Nuvion | mab | humanized | CD3 | Crohn's disease, ulcerative colitis | |
| Vobarilizumab[23] | scFv | humanized | IL6R | inflammatory autoimmune diseases | ||
| Volociximab[18] | mab | chimeric | integrin α5β1 | solid tumors | ||
| Vonlerolizumab | mab | humanized | CD134 | cancer | ||
| Vopratelimab[4] | mab | humanized | ICOS | ? | ||
| Vorsetuzumab mafodotin | mab | humanized | CD70 | cancer | ||
| Votumumab | HumaSPECT | mab | human | tumor antigen CTAA16.88 | colorectal tumors | |
| Vunakizumab[13] | mab | humanized | interleukin 17 alpha | ? | ||
| Xentuzumab[23] | mab | humanized | IGF1, IGF2 | ? | ||
| XMAB-5574 | ? | humanized | CD19 | diffuse large B-cell lymphoma | ||
| Zalutumumab[18] | HuMax-EGFr | mab | human | EGFR | squamous cell carcinoma of the head and neck | |
| Zanolimumab[5] | HuMax-CD4 | mab | human | CD4 | rheumatoid arthritis, psoriasis, T-cell lymphoma | |
| Zatuximab[33] | mab | chimeric | HER1 | cancer | ||
| Zenocutuzumab[6] | mab | humanized | ERBB3, HER3 | cancer | ||
| Ziralimumab[37] | mab | human | CD147 (basigin) | ? | ||
| Zolbetuximab[6] | mab | chimeric | CLDN18 | cancer | ||
| Zolimomab aritox | mab | mouse | CD5 | systemic lupus erythematosus, graft-versus-host disease |
References
- ↑ "United Therapeutics 10-K report". Retrieved 5 November 2011.
- 1 2 3 4 5 World Health Organization (2006). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 95" (PDF). WHO Drug Information. 20 (2).
- 1 2 3 4 5 6 World Health Organization (2013). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 109" (PDF). WHO Drug Information. 27 (2).
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 World Health Organization (2017). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 118" (PDF). WHO Drug Information. 31 (4).
- 1 2 3 4 5 6 7 World Health Organization (2004). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 90" (PDF). WHO Drug Information. 18 (1).
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 20 21 22 23 24 World Health Organization (2017). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 117" (PDF). WHO Drug Information. 31 (2).
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 World Health Organization (2015). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 113" (PDF). WHO Drug Information. 29 (2).
- 1 2 3 4 5 6 7 World Health Organization (2008). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 99" (PDF). WHO Drug Information. 22 (2).
- 1 2 3 4 5 6 7 8 9 World Health Organization (2007). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 98" (PDF). WHO Drug Information. 21 (4).
- 1 2 3 4 World Health Organization (2000). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 83" (PDF). WHO Drug Information. 14 (2).
- ↑ "Wyeth.com | Complete Project Listing". Wyeth. 2008. Archived from the original on June 12, 2008. Retrieved November 19, 2008.
- 1 2 3 4 5 6 7 8 9 World Health Organization (2002). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 87" (PDF). WHO Drug Information. 16 (2).
- 1 2 3 4 5 6 7 8 9 10 11 12 World Health Organization (2017). "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended INN: List 77" (PDF). WHO Drug Information. 31 (1).
- 1 2 3 World Health Organization (2003). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 88" (PDF). WHO Drug Information. 17 (1).
- 1 2 3 4 5 6 7 8 9 10 World Health Organization (2014). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 112" (PDF). WHO Drug Information. 28 (4).
- 1 2 3 4 5 6 World Health Organization (2010). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 104" (PDF). WHO Drug Information. 24 (4).
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 18 19 World Health Organization (2016). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 116" (PDF). WHO Drug Information. 30 (4).
- 1 2 3 4 5 6 7 8 World Health Organization (2008). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 93" (PDF). WHO Drug Information. 19 (2).
- 1 2 3 4 5 6 7 8 9 10 World Health Organization (2014). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 111" (PDF). WHO Drug Information. 28 (2).
- 1 2 3 4 World Health Organization (2004). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 92" (PDF). WHO Drug Information. 18 (4).
- 1 2 3 4 5 6 7 8 9 World Health Organization (2012). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 108" (PDF). WHO Drug Information. 26 (4).
- 1 2 World Health Organization (2013). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 110" (PDF). WHO Drug Information. 27 (4).
- 1 2 3 4 5 6 7 8 9 10 11 12 13 14 15 16 17 World Health Organization (2015). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 114" (PDF). WHO Drug Information. 29 (4).
- 1 2 3 4 5 World Health Organization (2011). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 105" (PDF). WHO Drug Information. 25 (2).
- 1 2 3 4 5 World Health Organization (2016). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 115" (PDF). WHO Drug Information. 30 (2).
- 1 2 3 4 World Health Organization (2007). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 97" (PDF). WHO Drug Information. 21 (2).
- 1 2 3 4 5 World Health Organization (2011). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 106" (PDF). WHO Drug Information. 25 (4).
- ↑ "BMS Shells Out $85M Up Front for Alder's Mid-Stage Rheumatoid Arthritis Antibody". Genetic Engineering & Biotechnology News. 10 November 2009.
- 1 2 World Health Organization (2009). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 101" (PDF). WHO Drug Information. 23 (2).
- 1 2 3 4 5 6 7 World Health Organization (2005). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 94" (PDF). WHO Drug Information. 19 (4).
- ↑ World Health Organization (1991). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 66" (PDF). WHO Drug Information. 5 (4).
- 1 2 3 World Health Organization (2018). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 119" (PDF). WHO Drug Information. 32 (2).
- 1 2 3 4 5 6 7 8 9 10 11 12 World Health Organization (2012). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 107" (PDF). WHO Drug Information. 26 (2).
- 1 2 3 4 World Health Organization (2001). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 85" (PDF). WHO Drug Information. 15 (2).
- ↑ World Health Organization (1997). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 77" (PDF). WHO Drug Information. 11 (2).
- ↑ World Health Organization (2004). "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended INN: List 51" (PDF). WHO Drug Information. 18 (1).
- 1 2 3 4 5 6 7 World Health Organization (2000). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 84" (PDF). WHO Drug Information. 14 (4).
- 1 2 3 4 World Health Organization (2004). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 91" (PDF). WHO Drug Information. 18 (2).
- 1 2 3 World Health Organization (2002). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 86" (PDF). WHO Drug Information. 16 (12).
- ↑ Buhmann, R; Simoes, B; Stanglmaier, M; Yang, T; Faltin, M; Bund, D; Lindhofer, H; Kolb, HJ; et al. (2008). "Immunotherapy of recurrent B–cell malignancies after allo SCT with Bi20 (FBTA05), a trifunctional anti-CD3 x anti-CD20 antibody and donor lymphocyte infusion". Bone Marrow Transplantation. 43 (5): 383–397. doi:10.1038/bmt.2008.323. PMID 18850012.
- ↑ Boehrer, S; Schroeder, Petra; Mueller, Tina; Atz, Judith; Chow, Kai Uwe; et al. (2011). "Cytotoxic effects of the trifunctional bispecific antibody FBTA05 in ex-vivo cells of chronic lymphocytic leukaemia depend on immune-mediated mechanisms". Anti-Cancer Drugs. 12 (10): 3085–3091. doi:10.1097/CAD.0b013e328344887f. PMID 21637160.
- ↑ Clinical trial number NCT00883896 for "Study to Evaluate the Safety and Efficacy of ILV-094 in Subjects With Rheumatoid Arthritis" at ClinicalTrials.gov
- ↑ Clinical trial number NCT00563524 for "Study Evaluating the Safety and Tolerability of ILV-094 in Subjects With Psoriasis" at ClinicalTrials.gov
- 1 2 3 4 World Health Organization (2011). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 103" (PDF). WHO Drug Information.
- ↑ http://www.prnewswire.com/news-releases/prometheus-obtains-exclusive-us-commercialization-rights-for-rencarex-121075044.html
- 1 2 World Health Organization (2009). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 102" (PDF). WHO Drug Information. 23 (4).
- ↑ NCI Drug Dictionary: Glemtumumab vedotin
- ↑ Clinical trial number NCT00537381 for "A Study of the Safety and Effectiveness of CNTO 95 in Patients With Metastatic Hormone Refractory Prostate Cancer" at ClinicalTrials.gov
- ↑ Clinical trial number NCT00246012 for "A Study of the Safety and Efficacy CNTO 95 in Subjects With Advanced Melanoma" at ClinicalTrials.gov
- 1 2 Guthrie, Arlo (27 June 2017). "Zoetis launches breakthrough treatment for canine atopic dermatitis". VetNurse News. Retrieved 2018-07-06.
- ↑ World Health Organization (2006). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 96" (PDF). WHO Drug Information. 20 (4).
- 1 2 3 4 5 World Health Organization (2008). "International Nonproprietary Names for Pharmaceutical Substances (INN). Proposed INN: List 100 (prepublication copy)" (PDF). WHO Drug Information.
- ↑ http://www.nejm.org/doi/full/10.1056/NEJMoa1606490
- 1 2 3 4 5 World Health Organization (2006). "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended INN: List 56" (PDF). WHO Drug Information. 20 (3).
- ↑ Xie, B.; Shen, J.; Dong, A.; Rashid, A.; Stoller, G.; Campochiaro, P. A. (2009). "Blockade of Sphingosine-1-phosphate Reduces Macrophage Influx and Retinal and Choroidal Neovascularization". Journal of Cellular Physiology. 218 (1): 192–198. doi:10.1002/jcp.21588. PMC 2905312. PMID 18781584.
- ↑ Jostein Dahle; Ada H. V. Repetto-Llamazares; Camilla S. Mollatt; Katrine B. Melhus; Oyvind S. Bruland; Arne Kolstad; Roy H. Larsen (January 2013). "Evaluating antigen targeting and anti-tumor activity of a new anti-CD37 radioimmunoconjugate against non-Hodgkin's lymphoma". Anticancer research. 33 (1): 85–95. PMID 23267131.
- ↑ Wright, Rob (13 February 2018). "Seattle Genetics Legacy Of Partnering". Life Science Leader. Beyond The Printed Page. Retrieved 22 February 2018.
- ↑ Ruf, P; Jäger, M; Ellwart, J; Wosch, S; Kusterer, E; Lindhofer, H; et al. (2004). "Two new trifunctional antibodies for the therapy of human malignant melanoma". International Journal of Cancer. 108 (5): 725–732. doi:10.1002/ijc.11630. PMID 14696099.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.