Indium(III) fluoride
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Indium(III) fluoride | |
| Other names
Indium trifluoride | |
| Identifiers | |
| ECHA InfoCard | 100.029.095 |
| EC Number | 232-005-0 |
PubChem CID |
|
| Properties | |
| InF3 | |
| Molar mass | 171.82 g/mol |
| Melting point | 1,172 °C (2,142 °F; 1,445 K)[1] |
| Structure | |
| Rhombohedral, hR24 | |
| R-3c, No. 167 | |
| Hazards | |
EU classification (DSD) (outdated) |
Irritant (Xi) |
| R-phrases (outdated) | R31, R36/37/38 |
| S-phrases (outdated) | S26, S36[2] |
| Flash point | non-flammable |
| Related compounds | |
Other anions |
Indium(III) chloride Indium(III) bromide Indium(III) iodide |
Other cations |
Aluminum fluoride Gallium(III) fluoride Thallium(I) fluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
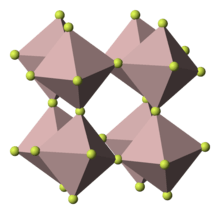
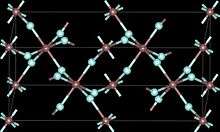
Indium(III) fluoride or indium trifluoride is the chemical compound composed of indium and fluorine with the formula InF3. It has a rhombohedral crystal structure identical to that of rhodium(III) fluoride .[3] It is formed by the reaction of indium(III) oxide with hydrogen fluoride or hydrofluoric acid.
Indium(III) fluoride is used in the synthesis of non-oxide glasses. It is also used as a catalyst for the addition of trimethylsilyl cyanide (TMSCN) to aldehydes to form cyanohydrins.[2]
References
- ↑ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, p. 112, ISBN 0-8493-0594-2, retrieved 2008-06-19
- 1 2 "435848 Indium(III) fluoride 99.9+ % trace metals basis". Sigma-Aldrich. Retrieved 2008-06-19.
- ↑ Fergusen, G.; Trotter, J. (1987), Structure Reports for 1984, Part A, Springer, p. 122, ISBN 90-277-2470-9, retrieved 2008-06-19
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.