Germanium tetrafluoride
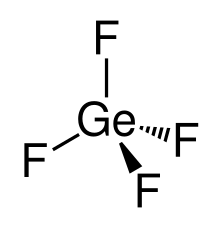 | |
| Names | |
|---|---|
| IUPAC names
Germanium tetrafluoride Tetrafluorogermane Tetrafluoridogermanium | |
| Other names
Germanium(IV) fluoride Germanium fluoride | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.029.101 |
| EC Number | 232-011-3 |
PubChem CID |
|
| |
| |
| Properties[1] | |
| GeF4 | |
| Molar mass | 148.634 g/mol |
| Appearance | colourless gas |
| Density | 6.074 g/L (gas), 2.46 g/mL (liquid)[2] |
| Melting point | −15 °C (5 °F; 258 K) at 4 bar |
| Boiling point | −36.5 °C (−33.7 °F; 236.7 K) sublimates |
| −50.0·10−6 cm3/mol | |
| Structure | |
| tetrahedral | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH |
-8.008 kJ/g |
| Hazards | |
| Main hazards | Reacts with water to form HF, corrosive |
| R-phrases (outdated) | R26 R35 |
| S-phrases (outdated) | S9 S26 S28 S36 S45 |
| NFPA 704 | |
| Flash point | Non-flammable |
| Related compounds | |
Other anions |
Germanium tetrachloride Germanium tetrabromide Germanium tetraiodide |
Other cations |
Carbon tetrafluoride Silicon tetrafluoride Tin tetrafluoride Lead tetrafluoride |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Germanium tetrafluoride (GeF4) is a chemical compound of germanium and fluorine. This colorless gas is formed by reacting germanium with fluorine or germanium dioxide (GeO2) with hydrofluoric acid (HF). Germanium difluoride can be synthesized by reacting germanium tetrafluoride with powdered germanium at 150–300 °C.[3]
Synthesis
Germanium tetrafluoride can be prepared by reaction of germanium with fluorine or hydrogen fluoride:
- Ge + 2 F2 → GeF4
It is also formed during the thermal decomposition of a complex salt, Ba[GeF6]:[4]
- Ba(GeF6) → GeF4 + BaF2
Properties
Germanium tetrafluoride is a noncombustible, strongly fuming gas with a garlic odor. It reacts with water to form hydrofluoric acid and germanium dioxide. Molecular decomposition occurs at temperatures above 1000 °C.[5]
Reaction of GeF4 with fluoride sources produces GeF5− anions with octahedral coordination around Ge atom due to polymerization.[6] The structural characterization of a discrete trigonal bipyramidal GeF5− anion was achieved by a novel "naked" fluoride reagent 1,3-bis(2,6-diisopropylphenyl)imidazolium fluoride.[7]
Uses
In combination with disilane, germanium tetrafluoride is used for in the synthesis of SiGe.[2]
References
- ↑ Lide, D. R., ed. (2005). CRC Handbook of Chemistry and Physics (86th ed.). Boca Raton (FL): CRC Press. p. 4.64. ISBN 0-8493-0486-5.
- 1 2 Germanium(IV) fluoride. sigmaaldrich.com
- ↑ Greenwood, N. N.; Earnshaw, A. (1998). Chemistry of the Elements (second edition). Butterworth Heinemann. pp. 376–377. ISBN 0-7506-3365-4.
- ↑ Georg Brauer: Handbuch der Präparativen Anorganischen Chemie
- ↑ Germaniumtetrafluorid. IFA Database
- ↑ Mallouk, T. E., Desbat, B., & Bartlett, N. (1984). Structural studies of salts of cis and trans .mu.-fluoro-bridged polymers of pentafluorogermanate(1-) and of the pentafluorogermanate(1-) monomer. Inorganic Chemistry, 23(20), 3160-3166. doi: 10.1021/ic00188a027
- ↑ Alič, B., Tramšek, M., Kokalj, A., & Tavčar, G. (2017). Discrete GeF5– Anion Structurally Characterized with a Readily Synthesized Imidazolium Based Naked Fluoride Reagent. Inorganic Chemistry, 56(16), 10070-10077. doi: 10.1021/acs.inorgchem.7b01606
