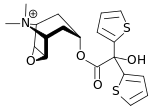
Tiotropium bromide
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Spiriva |
| MedlinePlus | a604018 |
| Pregnancy category |
|
| Routes of administration | Inhalation (oral) |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 19.5% (inhalation) |
| Metabolism |
Hepatic 25% (CYP2D6, CYP3A4) |
| Elimination half-life | 5–6 days |
| Excretion | Renal |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| ChEMBL | |
| ECHA InfoCard |
100.234.575 |
| Chemical and physical data | |
| Formula | C19H22BrNO4S2 |
| Molar mass | 472.416 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Tiotropium bromide, originally marketed as Spiriva,[1] is a long-acting, 24-hour, anticholinergic bronchodilator used in the management of chronic obstructive pulmonary disease (COPD).
Tiotroprium was discovered in 1991 by Boehringer Ingelheim and came to market in 2004.[2]
Medical uses
Tiotropium is used for maintenance treatment of chronic obstructive pulmonary disease (COPD) which includes chronic bronchitis and emphysema.[3] It is not however used for acute exacerbations.[3]
Adverse effects
Adverse effects are mainly related to its antimuscarinic effects. Common adverse drug reactions (≥1% of patients) associated with tiotropium therapy include: dry mouth and/or throat irritation. Rarely (<0.1% of patients) treatment is associated with:urinary retention, constipation, acute angle closure glaucoma, palpitations (notably supraventricular tachycardia and atrial fibrillation) and/or allergy (rash, angioedema, anaphylaxis).[4]
Tiotropium and another member of its class ipratropium were linked to increased risk of heart attacks, stroke and cardiovascular death.[5] The FDA requested further trials; these are now complete, and adequately resolve the previous safety concerns.[6]
Tiotropium mist inhaler (Respimat) has been found to be associated with an increase of all cause mortality in people with COPD.[7]
Mechanism of action
Tiotropium is a muscarinic receptor antagonist, often referred to as an antimuscarinic or anticholinergic agent. Although it does not display selectivity for specific muscarinic receptors, when topically applied it acts mainly on M3 muscarinic receptors[8] located on smooth muscle cells and submucosal glands. This leads to a reduction in smooth muscle contraction and mucus secretion and thus produces a bronchodilatory effect.
References
- ↑ "Tiotropium bromide". AdisInsight. Retrieved 8 March 2017.
- ↑ Corey, E.J. (2012). "Tiotropium bromide". Molecules and Medicine. John Wiley & Sons. ISBN 9781118361733.
- 1 2 "Spiriva Handihaler". The American Society of Health-System Pharmacists. Retrieved 3 April 2011.
- ↑ Rossi S, ed. (2006). Australian Medicines Handbook. Adelaide.
- ↑ Singh S, Loke YK, Furberg CD (September 2008). "Inhaled anticholinergics and risk of major adverse cardiovascular events in patients with chronic obstructive pulmonary disease: a systematic review and meta-analysis". JAMA. 300 (12): 1439–50. doi:10.1001/jama.300.12.1439. PMID 18812535.
- ↑ FDA. Follow-Up to the October 2008 Updated Early Communication about an Ongoing Safety Review of Tiotropium (marketed as Spiriva HandiHaler). FDA 2010
- ↑ Singh, S; Loke, YK; Enright, PL; Furberg, CD (Jun 14, 2011). "Mortality associated with tiotropium mist inhaler in patients with chronic obstructive pulmonary disease: systematic review and meta-analysis of randomised controlled trials". BMJ (Clinical research ed.). 342: d3215. doi:10.1136/bmj.d3215. PMC 3114950. PMID 21672999.
- ↑ Kato M, Komamura K, Kitakaze M (December 2006). "Tiotropium, a novel muscarinic M3 receptor antagonist, improved symptoms of chronic obstructive pulmonary disease complicated by chronic heart failure". Circ. J. 70 (12): 1658–60. doi:10.1253/circj.70.1658. PMID 17127817.