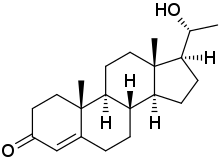
20β-Dihydroprogesterone
 | |
| Names | |
|---|---|
| IUPAC name
(8S,9S,10R,13S,14S,17S)-17-[(1R)-1-Hydroxyethyl]-10,13-dimethyl-1,2,6,7,8,9,11,12,14,15,16,17-dodecahydrocyclopenta[a]phenanthren-3-one | |
| Other names
20β-DHP; 20β-Hydroxyprogesterone; 20β-OHP; 20β-Hydroxypregn-4-en-3-one; Pregn-4-en-20β-ol-3-one | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C21H32O2 | |
| Molar mass | 316.485 g/mol |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
20β-Dihydroprogesterone (20β-DHP), also known as 20β-hydroxyprogesterone (20β-OHP), is an endogenous metabolite of progesterone which is formed by 20β-hydroxysteroid dehydrogenase.[1] It is a progestogen similarly to progesterone, with about one-fifth of the progestogenic activity of progesterone.[1]
See also
References
- 1 2 Bertram G. Katzung (30 November 2017). Basic and Clinical Pharmacology 14th Edition. McGraw-Hill Education. p. 728. ISBN 978-1-259-64116-9.
In addition to progesterone, 20α- and 20β-hydroxyprogesterone (20α- and 20β-hydroxy-4-pregnene-3-one) also are found. These compounds have about one-fifth the progestational activity of progesterone in humans and other species.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.