Alcohol oxidation
Alcohol oxidation is an important organic reaction. Primary alcohols (R-CH2-OH) can be oxidized either
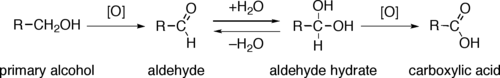
The indirect oxidation of primary alcohols to carboxylic acids normally proceeds via the corresponding aldehyde, which is transformed via an aldehyde hydrate (R-CH(OH)2) by reaction with water. The oxidation of a primary alcohol at the aldehyde level is possible by performing the reaction in absence of water, so that no aldehyde hydrate can be formed.
Oxidation to aldehydes
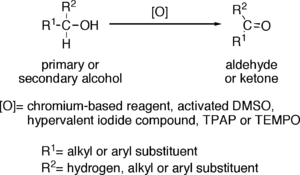
Oxidation of alcohols to aldehydes is partial oxidation; aldehydes are further oxidized to carboxylic acids. Conditions required for making aldehydes are heat and distillation. In aldehyde formation, the temperature of the reaction should be kept above the boiling point of the aldehyde and below the boiling point of the alcohol.
Reagents useful for the transformation of primary alcohols to aldehydes are normally also suitable for the oxidation of secondary alcohols to ketones. These include:
- Chromium-based reagents, such as Collins reagent (CrO3·Py2), PDC or PCC.
- Sulfonium species known as "activated DMSO" which can result from reaction of DMSO with electrophiles, such as oxalyl chloride (Swern oxidation), a carbodiimide (Pfitzner-Moffatt oxidation) or the complex SO3·Py (Parikh-Doering oxidation).
- Hypervalent iodine compounds, such as Dess-Martin periodinane or 2-Iodoxybenzoic acid.
- Catalytic TPAP in presence of excess of NMO (Ley oxidation).
- Catalytic TEMPO in presence of excess bleach (NaOCl) (Oxoammonium-catalyzed oxidation).
- Catalytic Redox-active copper complex in the presence of oxygen at room temperature.[1]
Allylic and benzylic alcohols can be oxidized in presence of other alcohols using certain selective oxidants such as manganese dioxide (MnO2).
Oxidation to ketones
Reagents useful for the oxidation of secondary alcohols to ketones, but normally inefficient for oxidation of primary alcohols to aldehydes, include chromium trioxide (CrO3) in a mixture of sulfuric acid and acetone (Jones oxidation) and certain ketones, such as cyclohexanone, in the presence of aluminium isopropoxide (Oppenauer oxidation). Another method is oxoammonium-catalyzed oxidation. Additionally, sodium hypochlorite (or household bleach) in acetone has been reported for efficient conversion of secondary alcohols in the presence of primary alcohols (Stevens oxidation).[2]
Oxidation to carboxylic acids
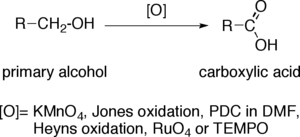
The direct oxidation of primary alcohols to carboxylic acids can be carried out using
- Potassium permanganate (KMnO4);
- Jones oxidation;
- PDC in DMF;
- Heyns oxidation;
- Ruthenium tetroxide (RuO4);
- or TEMPO.
Diol oxidation
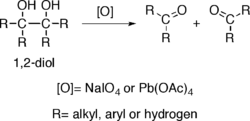
Alcohols possessing two hydroxy groups located on adjacent carbons —that is, 1,2-diols— suffer oxidative breakage at a carbon-carbon bond with some oxidants such as sodium periodate (NaIO4) or lead tetraacetate (Pb(OAc)4), resulting in generation of two carbonyl groups. The reaction is also known as glycol cleavage.
References
- Rajabimoghadam, Khashayar; Darwish, Yousef; Bashir, Umyeena; Pitman, Dylan; Eichelberger, Sidney; Siegler, Maxime A.; Swart, Marcel; Garcia-Bosch, Isaac (2018). "Catalytic Aerobic Oxidation of Alcohols by Copper Complexes Bearing Redox-Active Ligands with Tunable H-Bonding Groups". Journal of the American Chemical Society. 140 (48): 16625–16634. doi:10.1021/jacs.8b08748. PMC 6645702. PMID 30400740.
- Stevens, Robert; Chapman, Kevin T.; Weller, Harold N. (1980). "Convenient and inexpensive procedure for oxidation of secondary alcohols to ketones". Journal of Organic Chemistry. 45 (10): 2030–2032. doi:10.1021/jo01298a066.