Xanthan gum
 | |
| Names | |
|---|---|
| Other names
E 415 | |
| Identifiers | |
| ChemSpider |
|
| ECHA InfoCard | 100.031.255 |
| E number | E415 (thickeners, ...) |
| Properties | |
| C35H49O29 (monomer) | |
| Hazards | |
| R-phrases (outdated) | - |
| S-phrases (outdated) | - |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Xanthan gum (/ˈzænθən/) is a polysaccharide with many industrial uses, including as a common food additive. It is an effective thickening agent and stabilizer to prevent ingredients from separating. It can be produced from simple sugars using a fermentation process, and derives its name from the species of bacteria used, Xanthomonas campestris.
History
Xanthan gum was discovered by Allene Rosalind Jeanes and her research team at the United States Department of Agriculture, and brought into commercial production under the trade name Kelzan in the early 1960s.[2] It was approved for use in foods in 1968 and is accepted as a safe food additive in the USA, Canada, European countries, and many other countries, with E number E415.
Xanthan gum derives its name from the species of bacteria used during the fermentation process, Xanthomonas campestris.[3] This is the same bacterium responsible for causing black rot to form on broccoli, cauliflower, and other leafy vegetables.
Uses
Xanthan gum, 1%, can produce a significant increase in the viscosity of a liquid.[4]
In foods, xanthan gum is common in salad dressings and sauces. It helps to prevent oil separation by stabilizing the emulsion, although it is not an emulsifier. Xanthan gum also helps suspend solid particles, such as spices. Xanthan gum helps create the desired texture in many ice creams. Toothpaste often contains xanthan gum as a binder to keep the product uniform. Xanthan gum also helps thicken commercial egg substitutes made from egg whites, to replace the fat and emulsifiers found in yolks. It is also a preferred method of thickening liquids for those with swallowing disorders, since it does not change the color or flavor of foods or beverages at typical use levels.[5] In gluten-free baking xanthan gum is used to give the dough or batter the stickiness that would otherwise be achieved with gluten. In most foods, it is used at concentrations of 0.5% or less. Xanthan gum is used in wide range food products, such as sauces, dressings, meat and poultry products, bakery products, confectionery products, beverages, dairy products, others.
In the oil industry, xanthan gum is used in large quantities to thicken drilling mud.[6] These fluids serve to carry the solids cut by the drilling bit back to the surface. Xanthan gum provides great "low end" rheology. When the circulation stops, the solids still remain suspended in the drilling fluid. The widespread use of horizontal drilling and the demand for good control of drilled solids has led to its expanded use. It has also been added to concrete poured underwater, to increase its viscosity and prevent washout.
In cosmetics, xanthan gum is used to prepare water gels, usually in conjunction with bentonite clays. It is also used in oil-in-water emulsions to help stabilize the oil droplets against coalescence. It has some skin hydrating properties. Xanthan gum is a common ingredient in fake blood recipes, and in gunge/slime.
Shear thinning
The viscosity of xanthan gum solutions decreases with higher shear rates; this is called shear thinning or pseudoplasticity. This means that a product subjected to shear, whether from mixing, shaking or even chewing, will thin out, but, once the shear forces are removed, the food will thicken back up. In salad dressing, for example, the addition of xanthan gum makes it thick enough at rest in the bottle to keep the mixture fairly homogeneous, but the shear forces generated by shaking and pouring thins it, so it can be easily poured. When it exits the bottle, the shear forces are removed and it thickens again, so it clings to the salad.
Amounts used
The greater the ratio of xanthan gum added to a liquid, the thicker the liquid will become. An emulsion can be formed with as little as 0.1% (by weight). Increasing the amount of gum gives a thicker, more stable emulsion up to 1% xanthan gum. A teaspoon of xanthan gum weighs about 2.5 grams and brings one cup (250 ml) of water to a 1% concentration.[5][7]
To make a foam, 0.2–0.8% xanthan gum is typically used. Larger amounts result in larger bubbles and denser foam. Egg white powder (0.2–2.0%) with 0.1–0.4% xanthan gum yields bubbles similar to soap bubbles.
Health
Evaluation of workers exposed to xanthan gum dust found evidence of a link to respiratory symptoms.[8]
On May 20, 2011, the FDA issued a press release about SimplyThick, a food-thickening additive containing xanthan gum as the active ingredient, warning parents, caregivers and health care providers not to feed SimplyThick, a thickening product, to premature infants[9] The concern is that the product may cause premature infants to suffer necrotizing enterocolitis (NEC).
Allergies
The nutrients used in the production of xanthan gum may be derived from a variety of crop sources, some of which are known allergens, such as wheat, or soy.
In fermentation processes, such as in the production of xanthan gum, nutrients are needed as a food source by the fermentation organism. Typical nutrients used in fermentation processes are: soybean proteins, and maize (corn) or wheat starches. However, products made by fermentation, such as gums, are not composed of the source nutrients, these are metabolized during fermentation. Then, after the fermentation step, the gums are recovered and purified, dried and milled.[10] Nutrients are not transferred directly to the final products, although unavoidable residues of the process may be present.
Due to the presence of low levels of residual protein material or protein fragments in xanthan gum it is possible that in isolated cases, an adverse reaction to these proteins, such as allergy or sensitization, could result. Instances of this occurring are, however, extremely rare[11] and xanthan gum is not listed as a known allergen or sensitizer for the general public.[12][13][14] In fact, xanthan gum is notably used to replace grains or other sensitizers in products intended for individuals with sensitivities or allergies to grains.
Xanthan gum is a "highly efficient laxative," according to a study that fed 15 g/day for 10 days to 18 normal volunteers. This is not a dosage that would be encountered in normal consumption of foodstuffs. This study set out to examine the use of xanthan gum as a laxative. As described above, xanthan gum binds water very efficiently, which significantly aids passing stools.[15] Some people react to much smaller amounts of xanthan gum with symptoms of intestinal bloating and diarrhea.
There are many substitutes for xanthan gum when used for baking such as guar gum and locust bean gum.
Preparation
Xanthan gum is produced by the fermentation of glucose, sucrose, or lactose.
The polysaccharide is prepared by the bacteria being inoculated into a sterile aqueous solution of carbohydrate(s), a source of nitrogen, dipotassium phosphate, and some trace elements. The medium is well-aerated and stirred, and the xanthan polymer is produced extracellularly into the medium. After one to four days, the polymer is precipitated from the medium by the addition of isopropyl alcohol, and the precipitate is dried and milled to give a powder that is readily soluble in water or brine.
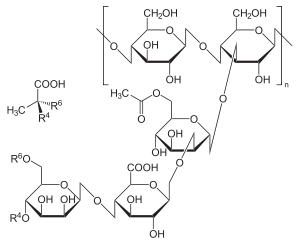
It is composed of pentasaccharide repeat units, comprising glucose, mannose, and glucuronic acid in the molar ratio 2:2:1.[16]
A strain of X. campestris has been developed that will grow on lactose - which allows it to be used to process whey, a waste product of cheese production. This can produce 30 g/L of xanthan gum for every 40 g/L of whey powder. Whey-derived xanthan gum is commonly used in many commercial products, such as shampoos and salad dressings.[17]
Detail of the biosynthesis
Synthesis originates from glucose as substrate for synthesis of the sugar nucleotides precursors UDP-glucose, UDP-glucuronate, and GDP-mannose that are required for building the pentasaccharide repeat unit. This links the synthesis of xanthan to the central carbohydrate metabolism. The repeat units are built up at undecaprenylphosphate lipid carriers that are anchored in the cytoplasmic membrane.
Specific glycosyltransferases sequentially transfer the sugar moieties of the nucleotide sugar xanthan precursors to the lipid carriers. Acetyl and pyruvyl residues are added as non-carbohydrate decorations. Mature repeat units are polymerized and exported in a way resembling the Wzy-dependent polysaccharide synthesis mechanism of Enterobacteriaceae. Products of the gum gene cluster drive synthesis, polymerization, and export of the repeat unit.[18]
References
- ↑ "Sicherheitsdatenblatt des Herstellers Carl-Roth" (PDF). Archived (PDF) from the original on 2011-07-18. Retrieved 2011-04-18.
- ↑ Whistler, Roy, L, and BeMiller, James N., eds Industrial Gums: Polysaccharides and their Derivatives Academic Press (1973) ISBN 0-12-746252-X.
- ↑ Barrére, G.C., C.E. Barber, and M.J. Daniels (1986) Intl. J. Biological Macromolecules, 8(6):372-374
- ↑ Davidson, Robert L. (1980). Handbook of Water-soluble Gums and Resins. McGraw Hill. ISBN 0-07-015471-6.
- 1 2 cuisine, m. (2014). Xanthan Gum. Retrieved from modernist cuisine: "Archived copy". Archived from the original on 2014-06-18. Retrieved 2014-06-21.
- ↑ "Oilfield Glossary - xanthan gum". www.glossary.oilfield.slb.com. Schlumberger. Archived from the original on 12 February 2017. Retrieved 30 April 2017.
- ↑ Tests and measurements of xanthan gum "Archived copy". Archived from the original on 2016-03-04. Retrieved 2016-01-02.
- ↑ Sargent, EV.; Adolph, J.; Clemmons, MK.; Kirk, GD.; Pena, BM.; Fedoruk, MJ. (Jul 1990). "Evaluation of flu-like symptoms in workers handling xanthan gum powder". J Occup Med. 32 (7): 625–30. doi:10.1097/00043764-199007000-00014. PMID 2391577.
- ↑ {{cite web |url=http://www.simplythick.com/Safety |title=SimplyThick Warning
- ↑ Xanthan gum production, recovery and properties. F Garcia-Ochoa et al., Biotechnol Advances, (2000), 18, 549.
- ↑ Ingredient and labeling issues associated with allergenic foods. S. L. Taylor, S. L. Hefle, Allergy 2001: 56: Suppl. 67: 64–69
- ↑ 2. European Food Safety Authority Scientific Opinion on the Evaluation of Allergenic Foods and Food Ingredients for Labelling Purposes, EFSA Journal 2014, 12(11), 3894.
- ↑ United States Food Allergen Labeling and Consumer Protection Act of 2004 (FALCPA).
- ↑ Dietary effects of xanthan gum in man. M.A. Eastwood et al., Food Additives & Contams. (1987), 4(1), 17.
- ↑ Daly, J.; Tomlin, J.; Read, NW. (May 1993). "The effect of feeding xanthan gum on colonic function in man: correlation with in vitro determinants of bacterial breakdown". Br J Nutr. 69 (3): 897–902. doi:10.1079/BJN19930089. PMID 8329363.
- ↑ Garcı́a-Ochoa, F; Santos, V.E; Casas, J.A; Gómez, E (2000). "Xanthan gum: production, recovery, and properties". Biotechnology Advances. 18 (7): 549–579. doi:10.1016/S0734-9750(00)00050-1. ISSN 0734-9750.
- ↑ Tortora, G.J., Funke, B.R., & Case, C.L. (2010). Microbiology: An Introduction, 10th edition. San Francisco: Benjamin Cummings. Pg. 801.
- ↑ Becker and Vorholter (2009). "Xanthan Biosynthesis by Xanthomonas Bacteria: An Overview of the Current Biochemical and Genomic Data". Microbial Production of Biopolymers and Polymer Precursors. Caister Academic Press. ISBN 978-1-904455-36-3.