Spiropentane
 | |
| Names | |
|---|---|
| Preferred IUPAC name
Spiro[2.2]pentane | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| C5H8 | |
| Molar mass | 68.12 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Spiropentane is a hydrocarbon with formula C5H8. It is the simplest spiro-connected cycloalkane, a triangulane.[1][2][3][4] It took several years after the discovery in 1887 until the structure of the molecule was determined.[5][6][7]According to the nomenclature rules for spiro compounds, the systematic name is spiro[2.2]pentane. However, there can be no constitutive isomeric spiropentanes, hence the name is unique without brackets and numbers.
Synthesis
After Gustavson produced cyclopropane by reacting 1,3-dibromopropane with dust fine zinc, he tried the same reaction with 2,2-Bis(bromomethyl)-1,3-dibromopropane (see formula scheme). The starting material is easily obtained by reacting pentaerythritol with hydrobromic acid. A molecule with the formula C5H8 was obtained. It was referred to as vinyltrimethylene in the initial publication.[8] In 1907, Fecht expressed the assumption that it must be spiropentane, a constitutional isomer of vinylcyclopropane.[9]Further Evidence for the structure of the hydrocarbon comes from the fact that it could also be obtained from 1,1-bis(bromomethyl)-cyclopropane (see formula scheme).[10]
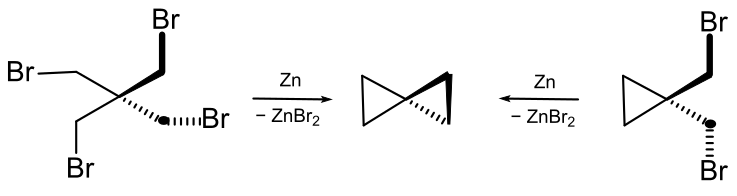
Spiropentane is difficult to separate from the other reaction products and the early procedures resulted in impure mixtures. Decades later, the production method was improved. The spiro hydrocarbon can be separated from the byproducts (2-methyl-1-butene, 1,1-dimethylcyclopropane, methylenecyclobutane) by distillation.[11]
References
- ↑ Donohue, Jerry; Humphrey, George L.; Schomaker, Verner (1945). "The Structure of Spiropentane". Journal of the American Chemical Society. 67 (2): 332–335. doi:10.1021/ja01218a056. ISSN 0002-7863.
- ↑ Murray, M. J.; Stevenson, Eugene H. (1944). "SPIROPENTANE". Journal of the American Chemical Society. 66 (2): 314–314. doi:10.1021/ja01230a515. ISSN 0002-7863.
- ↑ Murray, M. J.; Stevenson, Eugene H. (1944). "The Debromination of Pentaerythrityl Bromide by Zinc. Isolation of Spiropentane1". Journal of the American Chemical Society. 66 (5): 812–816. doi:10.1021/ja01233a047. ISSN 0002-7863.
- ↑ Price, J.E.; Coulterpark, K.A.; Masiello, T.; Nibler, J.W.; Weber, A.; Maki, A.; Blake, T.A. (2011). "High-resolution infrared spectra of spiropentane, C5H8". Journal of Molecular Spectroscopy. 269 (1): 129–136. doi:10.1016/j.jms.2011.05.011. ISSN 0022-2852.
- ↑ Philipow, O. (1916). "Die Konstitution der Kohlenwasserstoffe Gustavsons: Vinyltrimethylen und Äthylidentrimethylen". Journal für Praktische Chemie. 93 (1): 162–182. doi:10.1002/prac.19160930112. ISSN 0021-8383.
- ↑ Faworsky, Al.; Batalin, W. (1914). "Über das Vinyltrimethylen und Äthyliden-trimethylen von Gustavson". Berichte der deutschen chemischen Gesellschaft. 47 (2): 1648–1651. doi:10.1002/cber.19140470250. ISSN 0365-9496.
- ↑ Burns, G.R.; McGavin, D.G., Infrared and Raman spectra of spiropentane-H8, Applied Spectroscopy 26(5):540–542, 1 September 1972.
- ↑ Gustavson, G. (1896). "Ueber Aethylidentrimethylen". Journal für Praktische Chemie. 54 (1): 104–107. doi:10.1002/prac.18960540106. ISSN 0021-8383.
- ↑ Fecht, H. (1907). "Über Spirocyclane". Berichte der deutschen chemischen Gesellschaft. 40 (3): 3883–3891. doi:10.1002/cber.190704003194. ISSN 0365-9496.
- ↑ Zelinsky, N. (1913). "Über das Spirocyclan, seine Synthese und sein Verhalten bei der Reduktionskatalyse". Berichte der deutschen chemischen Gesellschaft. 46 (1): 160–172. doi:10.1002/cber.19130460128. ISSN 0365-9496.
- ↑ Applequist, Douglas E.; Fanta, George F.; Henrikson, Bertel W. (1958). "Chemistry of Spiropentane. I. An Improved Synthesis of Spiropentane". The Journal of Organic Chemistry. 23 (11): 1715–1716. doi:10.1021/jo01105a037. ISSN 0022-3263.