Platinum disulfide
 | |
| Names | |
|---|---|
| Other names
platinum(IV) sulfide, platinic sulfide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| EC Number | 234-876-2 |
PubChem CID |
|
| |
| |
| Properties | |
| PtS2 | |
| Molar mass | 259.20 g·mol−1 |
| Appearance | black solid |
| Density | 7.86 g/cm3 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
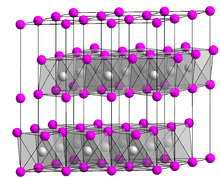
Platinum disulfide is the inorganic compound with the formula PtS2. It is a black, semiconducting solid, which is insoluble in all solvents. The compound adopts the cadmium iodide structure, being composed of sheets of octahedral Pt and pyramidal sulfide centers. Single crystals are grown by chemical vapor transport using phosphorus as the transport agent.[1] A related compound is platinum(II) sulfide, PtS.
References
- ↑ S. Soled, A. Wold (1979). "Platinum Disulfide and Platinum Ditelluride". Inorganic Syntheses. 19: 49–51. doi:10.1002/9780470132500.ch9.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.