Terpenoid
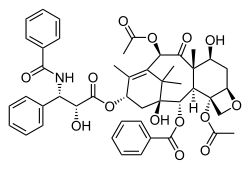
The terpenoids (/ˈtɜːrpɪnɔɪd/ TUR-pin-oyd), sometimes called isoprenoids, are a large and diverse class of naturally occurring organic chemicals derived from terpenes. Most are multicyclic structures with oxygen-containing functional groups. About 60% of known natural products are terpenoids.[1] Although sometimes used interchangeably with "terpenes", terpenoids contain additional functional groups, usually O-containing.[2] Terpenes are hydrocarbons.
Plant terpenoids are used for their aromatic qualities and play a role in traditional herbal remedies. Terpenoids contribute to the scent of eucalyptus, the flavors of cinnamon, cloves, and ginger, the yellow color in sunflowers, and the red color in tomatoes.[3] Well-known terpenoids include citral, menthol, camphor, salvinorin A in the plant Salvia divinorum, the cannabinoids found in cannabis, ginkgolide and bilobalide found in Ginkgo biloba, and the curcuminoids found in turmeric and mustard seed.
The steroids and sterols in animals are biologically produced from terpenoid precursors. Sometimes terpenoids are added to proteins, e.g., to enhance their attachment to the cell membrane; this is known as isoprenylation.
-Humulone.svg.png)
Structure and classification
Terpenoids are modified terpenes, wherein methyl groups have been moved or removed, or oxygen atoms added. (Some authors use the term "terpene" more broadly, to include the terpenoids.) Just like terpenes, the terpenoids can be classified according to the number of isoprene units that comprise the parent terpene:
- Hemiterpenoids, 1 isoprene unit (5 carbons)
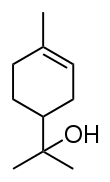
- Monoterpenoids, 2 isoprene units (10C, i.e., derived from monoterpenes)
- Sesquiterpenoids, 3 isoprene units (15C, i.e., derived from sesquiterpenes)
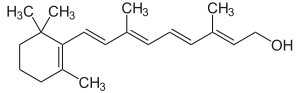
- Diterpenoids, 4 isoprene units (20C, i.e., derived from diterpenes). ginkgolides.
- Sesterterpenoids, 5 isoprene units (25C, i.e., derived from sesterterpenes).
- Triterpenoids, 6 isoprene units (30C, i.e., derived from triterpenes). Example: sterols.
- Tetraterpenoids, 8 isoprene units (40C, i.e., derived from tetraterpenes). Example: carotenoids)
- Polyterpenoid with a larger number of isoprene units
Terpenoids can also be classified according to the number of cyclic structures they contain. The Salkowski test can be used to identify the presence of terpenoids.[4]
Biosynthesis
Terpenoids, at least those containing an alcohol functional group, often arise by hydrolysis of carbocationic intermediates produced from geranyl pyrophosphate. Analogously hydrolysis of intermediates from farnesyl pyrophosphate gives sesquiterpenoids, and hydrolysis of intermediates from geranylgeranyl pyrophosphate gives diterpenoids, etc.[5]
See also
References
- ↑ Firn R (2010). Nature's Chemicals. Oxford: Biology.
- ↑ https://goldbook.iupac.org/html/T/T06279.html
- ↑ Specter M (September 28, 2009). "A Life of Its Own". The New Yorker.
- ↑ Ayoola GA (2008). "Phytochemical Screening and Antioxidant Activities of Some Selected Medicinal Plants Used for Malaria Therapy in Southwestern Nigeria". Tropical Journal of Pharmaceutical Research. 7 (3): 1019–1024. doi:10.4314/tjpr.v7i3.14686.
- ↑ Davis, Edward M.; Croteau, Rodney (2000). "Cyclization Enzymes in the Biosynthesis of Monoterpenes, Sesquiterpenes, and Diterpenes". Topics in Current Chemistry. 209: 53–95. doi:10.1007/3-540-48146-X_2.