Geranyl pyrophosphate
 | |
 | |
| Identifiers | |
|---|---|
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| MeSH | Geranyl+pyrophosphate |
PubChem CID |
|
| |
| |
| Properties | |
| C10H20O7P2 | |
| Molar mass | 314.209 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Geranyl pyrophosphate (GPP), also known as geranyl diphosphate (GDP), is an intermediate in the HMG-CoA reductase pathway used by organisms in the biosynthesis of farnesyl pyrophosphate, geranylgeranyl pyrophosphate.[1] These species are respectively precursors to sesquiterpenes and diterpenes.[2]
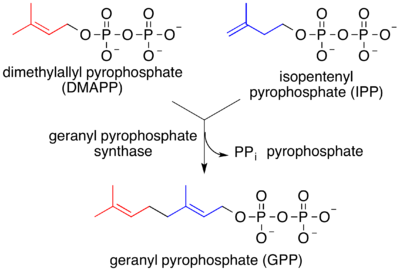
Isopentenyl pyrophosphate (IPP) and dimethylallyl pyrophosphate (DMAPP) are condensed by geranyl pyrophosphate synthase (dimethylallyltranstransferase) to produce geranyl pyrophosphate (GPP) and pyrophosphate. The carbon skeletons of DMAPP and IPP have been colored to indicate their location in GPP.
Related compounds
See also
Further reading
- Kulkarni RS, Pandit SS, Chidley HG, Nagel R, Schmidt A, Gershenzon J, Pujari KH, Giri AP and Gupta VS, 2013, Characterization of three novel isoprenyl diphosphate synthases from the terpenoid rich mango fruit. Plant Physiology and Biochemistry, 71, 121–131.
References
- ↑ Davis, Edward M.; Croteau, Rodney (2000). "Cyclization Enzymes in the Biosynthesis of Monoterpenes, Sesquiterpenes, and Diterpenes". Topics in Current Chemistry. 209: 53–95. doi:10.1007/3-540-48146-X_2.
- ↑ Bohlmann, Jorg; Gershenzon, Jonathan (2009). "Old Substrates for New Enzymes of Terpenoid Biosynthesis". Proceedings of the National Academy of Sciences of the United States of America. 106: 10402–10403. doi:10.1073/pnas.0905226106. PMC 2705528.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.