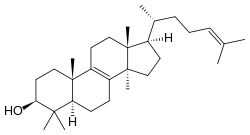
Lanosterol
 | |
 | |
| Names | |
|---|---|
| IUPAC name
lanosta-8,24-dien-3-ol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.105 |
| MeSH | Lanosterol |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C30H50O | |
| Molar mass | 426.71 g/mol |
| Melting point | 138 to 140 °C (280 to 284 °F; 411 to 413 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Lanosterol is a tetracyclic triterpenoid and is the compound from which all animal and fungal steroids are derived. By contrast plant steroids are produced via cycloartenol.[1]
Role in creation of steroids
Elaboration of lanosterol under enzyme catalysis leads to the core structure of steroids. 14-Demethylation of lanosterol by CYP51 eventually yields cholesterol.
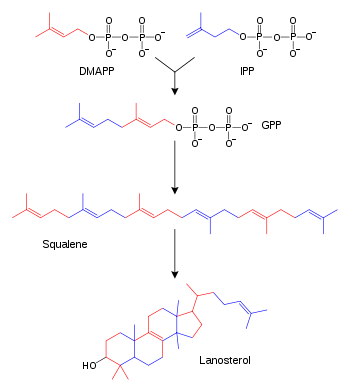
Simplified version of the lanosterol synthesis pathway with the intermediates isopentenyl pyrophosphate (IPP), dimethylallyl pyrophosphate (DMAPP), geranyl pyrophosphate (GPP), and squalene shown. Some intermediates are omitted.
Recent research suggests that lanosterol might be instrumental in prevention of formation of cataracts in mammals.[2]
Biosynthesis
| Description | Illustration | Enzyme |
| Two molecules of farnesyl pyrophosphate condense with reduction by NADPH to form squalene | squalene synthase | |
| Squalene is oxidized to 2,3-oxidosqualene (squalene epoxide) | 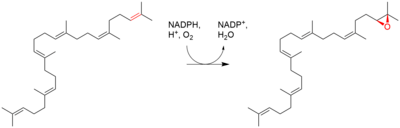 | squalene monooxygenase |
| 2,3-Oxidosqualene is converted to a protosterol cation and finally to lanosterol |  | lanosterol synthase |
| (step 2) | 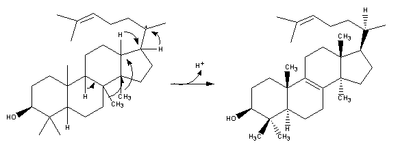 | (step 2) |
See also
References
- ↑ Schaller, Hubert (May 2003). "The role of sterols in plant growth and development". Progress in Lipid Research. 42 (3): 163–175. doi:10.1016/S0163-7827(02)00047-4.
- ↑ Ling Zhao; Xiang-Jun Chen; Jie Zhu; Yi-Bo Xi; Xu Yang; Li-Dan Hu; Hong Ouyang; Sherrina H. Patel; Xin Jin; Danni Lin; Frances Wu; Ken Flagg; Huimin Cai; Gen Li; Guiqun Cao; Ying Lin; Daniel Chen; Cindy Wen; Christopher Chung; Yandong Wang; Austin Qiu; Emily Yeh; Wenqiu Wang; Xun Hu; Seanna Grob; et al. (July 2015). "Lanosterol reverses protein aggregation in cataracts". Nature. doi:10.1038/nature14650.
- E. J. Corey, W. E. Russey, P. R. Ortiz de Montellano (1966). "2,3-Oxidosqualene, an Intermediate in the Biological Synthesis of Sterols from Squalene". Journal of the American Chemical Society. 88 (20): 4750–4751. doi:10.1021/ja00972a056. PMID 5918046.
- I. Abe; M. Rohmer; G. D. Prestwich (1993). "Enzymatic cyclization of squalene and oxidosqualene to sterols and triterpenes". Chemical Reviews. 93 (6): 2189–2206. doi:10.1021/cr00022a009.
- A. Eschenmoser, L. Ruzicka, O. Jeger, D. Arigoni (1955). "Zur Kenntnis der Triterpene. 190. Mitteilung. Eine stereochemische Interpretation der biogenetischen Isoprenregel bei den Triterpenen". Helvetica Chimica Acta. 38 (7): 1890–1904. doi:10.1002/hlca.19550380728.
External links
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.