Sirtuin
Sirtuins are a class of proteins that possess either mono-ADP-ribosyltransferase, or deacylase activity, including deacetylase, desuccinylase, demalonylase, demyristoylase and depalmitoylase activity.[2][3][4] The name Sir2 comes from the yeast gene 'silent mating-type information regulation 2',[5] the gene responsible for cellular regulation in yeast.
| Sir2 family | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
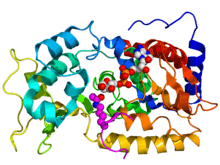 Crystallographic structure of yeast sir2 (rainbow colored cartoon, N-terminus = blue, C-terminus = red) complexed with ADP (space-filling model, carbon = white, oxygen = red, nitrogen = blue, phosphorus = orange) and a histone H4 peptide (magenta) containing an acylated lysine residue (displayed as spheres).[1] | |||||||||||
| Identifiers | |||||||||||
| Symbol | SIR2 | ||||||||||
| Pfam | PF02146 | ||||||||||
| Pfam clan | CL0085 | ||||||||||
| InterPro | IPR003000 | ||||||||||
| PROSITE | PS50305 | ||||||||||
| SCOPe | 1j8f / SUPFAM | ||||||||||
| |||||||||||
From in vitro studies, sirtuins are implicated in influencing cellular processes like aging, transcription, apoptosis, inflammation[6] and stress resistance, as well as energy efficiency and alertness during low-calorie situations.[7] As of 2018, there was no clinical evidence that sirtuins affect human aging.[8]
Yeast Sir2 and some, but not all, sirtuins are protein deacetylases. Unlike other known protein deacetylases, which simply hydrolyze acetyl-lysine residues, the sirtuin-mediated deacetylation reaction couples lysine deacetylation to NAD hydrolysis. This hydrolysis yields O-acetyl-ADP-ribose, the deacetylated substrate and nicotinamide, which is an inhibitor of sirtuin activity itself. The dependence of sirtuins on NAD+ links their enzymatic activity directly to the energy status of the cell via the cellular NAD+:NADH ratio, the absolute levels of NAD+, NADH or nicotinamide or a combination of these variables.
Sirtuins that deacetylate histones are structurally and mechanistically distinct from other classes of histone deacetylases (classes I, IIA, IIB and IV), which have a different protein fold and use Zn2+ as a cofactor.[9][10]
Actions and species distribution
Sirtuins are a family of signaling proteins involved in metabolic regulation.[11][12] They are ancient in animal evolution and appear to possess a highly conserved structure throughout all kingdoms of life.[11] Whereas bacteria and archaea encode either one or two sirtuins, eukaryotes encode several sirtuins in their genomes. In yeast, roundworms, and fruitflies, sir2 is the name of one of the sirtuin-type proteins (see table below).[13] Research on sirtuin protein was started in 1991 by Leonard Guarente of MIT.[14][15] Mammals possess seven sirtuins (SIRT1–7) that occupy different subcellular compartments: SIRT1, SIRT6 and SIRT7 are predominantly in the nucleus, SIRT2 in the cytoplasm, and SIRT3, SIRT4 and SIRT5 in the mitochondria.[11]
Types
The first sirtuin was identified in yeast (a lower eukaryote) and named sir2. In more complex mammals, there are seven known enzymes that act in cellular regulation, as sir2 does in yeast. These genes are designated as belonging to different classes (I-IV), depending on their amino acid sequence structure.[16] Several gram positive prokaryotes as well as the gram negative hyperthermophilic bacterium Thermotoga maritima possess sirtuins that are intermediate in sequence between classes, and these are placed in the "undifferentiated" or "U" class. In addition, several Gram positive bacteria, including Staphylococcus aureus and Streptococcus pyogenes, as well as several fungi carry macrodomain-linked sirtuins (termed "class M" sirtuins).[4]
| Class | Subclass | Species | Intracellular location |
Activity | Function | |||
|---|---|---|---|---|---|---|---|---|
| Bacteria | Yeast | Mouse | Human | |||||
| I | a | Sir2 or Sir2p, Hst1 or Hst1p | Sirt1 | SIRT1 | Nucleus, cytoplasm | Deacetylase | Metabolism inflammation | |
| b | Hst2 or Hst2p | Sirt2 | SIRT2 | Nucleus and cytoplasm | Deacetylase | Cell cycle, tumorigenesis | ||
| Sirt3 | SIRT3 | Mitochondria | Deacetylase | Metabolism | ||||
| c | Hst3 or Hst3p, Hst4 or Hst4p | |||||||
| II | Sirt4 | SIRT4 | Mitochondria | ADP-ribosyl transferase | Insulin secretion | |||
| III | Sirt5 | SIRT5 | Mitochondria | Demalonylase, desuccinylase and deacetylase | Ammonia detoxification | |||
| IV | a | Sirt6 | SIRT6 | Nucleus | Demyristoylase, depalmitoylase, ADP-ribosyl transferase and deacetylase | DNA repair, metabolism, TNF secretion | ||
| b | Sirt7 | SIRT7 | Nucleolus | Deacetylase | rRNA transcription | |||
| U | cobB[17] | Regulation of acetyl-CoA synthetase[18] | metabolism | |||||
| M | SirTM[4] | ADP-ribosyl transferase | ROS detoxification | |||||
SIRT3, a mitochondrial protein deacetylase, plays a role in the regulation of multiple metabolic proteins like isocitrate dehydrogenase of the TCA cycle. It also plays a role in skeletal muscle as a metabolic adaptive response. Since glutamine is a source of a-ketoglutarate used to replenish the TCA cycle, SIRT4 is involved in glutamine metabolism.[19]
Aging
Although preliminary studies with resveratrol, an activator of deacetylases such as SIRT1,[20] led some scientists to speculate that resveratrol may extend lifespan, there was no clinical evidence for such an effect, as of 2018.[8]
In vitro studies shown that calorie restriction regulates the plasma membrane redox system, involved in mitochondrial homeostasis, and the reduction of inflammation through cross-talks between SIRT1 and AMP-activated protein kinase (AMPK),[21][22][23] but the role of sirtuins in longevity is still unclear,[20][21][23] as calorie restriction in yeast could extend lifespan in the absence of Sir2 or other sirtuins, while the in vivo activation of Sir2 by calorie restriction or resveratrol to extend lifespan has been challenged in multiple organisms.[24]
Tissue fibrosis
A 2018 review indicated that SIRT levels are lower in tissues from people with scleroderma, and such reduced SIRT levels may increase risk of fibrosis through modulation of the TGF-β signaling pathway.[25]
DNA repair
SIRT1, SIRT6 and SIRT7 proteins are employed in DNA repair.[26] SIRT1 protein promotes homologous recombination in human cells and is involved in recombinational repair of DNA breaks.[27]
SIRT6 is a chromatin-associated protein and in mammalian cells is required for base excision repair of DNA damage.[28] SIRT6 deficiency in mice leads to a degenerative aging-like phenotype.[28] In addition, SIRT6 promotes the repair of DNA double-strand breaks.[29] Furthermore, over-expression of SIRT6 can stimulate homologous recombinational repair.[30]
SIRT7 knockout mice display features of premature aging.[31] SIRT7 protein is required for repair of double-strand breaks by non-homologous end joining.[31]
Inhibitors
Sirtuin activity is inhibited by nicotinamide, which binds to a specific receptor site.[32]
References
- PDB: 1szd; Zhao K, Harshaw R, Chai X, Marmorstein R (June 2004). "Structural basis for nicotinamide cleavage and ADP-ribose transfer by NAD(+)-dependent Sir2 histone/protein deacetylases". Proceedings of the National Academy of Sciences of the United States of America. 101 (23): 8563–8. Bibcode:2004PNAS..101.8563Z. doi:10.1073/pnas.0401057101. PMC 423234. PMID 15150415.
- Du J, Zhou Y, Su X, Yu JJ, Khan S, Jiang H, Kim J, Woo J, Kim JH, Choi BH, He B, Chen W, Zhang S, Cerione RA, Auwerx J, Hao Q, Lin H (November 2011). "Sirt5 is a NAD-dependent protein lysine demalonylase and desuccinylase". Science. 334 (6057): 806–9. Bibcode:2011Sci...334..806D. doi:10.1126/science.1207861. PMC 3217313. PMID 22076378.
- Jiang H, Khan S, Wang Y, Charron G, He B, Sebastian C, Du J, Kim R, Ge E, Mostoslavsky R, Hang HC, Hao Q, Lin H (April 2013). "SIRT6 regulates TNF-α secretion through hydrolysis of long-chain fatty acyl lysine". Nature. 496 (7443): 110–3. Bibcode:2013Natur.496..110J. doi:10.1038/nature12038. PMC 3635073. PMID 23552949.
- Rack JG, Morra R, Barkauskaite E, Kraehenbuehl R, Ariza A, Qu Y, Ortmayer M, Leidecker O, Cameron DR, Matic I, Peleg AY, Leys D, Traven A, Ahel I (July 2015). "Identification of a Class of Protein ADP-Ribosylating Sirtuins in Microbial Pathogens". Molecular Cell. 59 (2): 309–20. doi:10.1016/j.molcel.2015.06.013. PMC 4518038. PMID 26166706.
- EntrezGene 23410
- Preyat N, Leo O (May 2013). "Sirtuin deacylases: a molecular link between metabolism and immunity". Journal of Leukocyte Biology. 93 (5): 669–80. doi:10.1189/jlb.1112557. PMID 23325925.
- Satoh A, Brace CS, Ben-Josef G, West T, Wozniak DF, Holtzman DM, Herzog ED, Imai S (July 2010). "SIRT1 promotes the central adaptive response to diet restriction through activation of the dorsomedial and lateral nuclei of the hypothalamus". The Journal of Neuroscience. 30 (30): 10220–32. doi:10.1523/JNEUROSCI.1385-10.2010. PMC 2922851. PMID 20668205.
- Shetty, Ashok K.; Kodali, Maheedhar; Upadhya, Raghavendra; Madhu, Leelavathi N. (2018). "Emerging anti-aging strategies - scientific basis and efficacy (Review)". Aging and Disease. 9 (6): 1165–1184. doi:10.14336/ad.2018.1026. ISSN 2152-5250. PMC 6284760. PMID 30574426.
- Bürger M, Chory J (2018). "Structural and chemical biology of deacetylases for carbohydrates, proteins, small molecules and histones". Communications Biology. 1: 217. doi:10.1038/s42003-018-0214-4. PMC 6281622. PMID 30534609.
- Marks PA, Xu WS (July 2009). "Histone deacetylase inhibitors: Potential in cancer therapy". Journal of Cellular Biochemistry. 107 (4): 600–8. doi:10.1002/jcb.22185. PMC 2766855. PMID 19459166.
- Ye, X; Li, M; Hou, T; Gao, T; Zhu, WG; Yang, Y (3 January 2017). "Sirtuins in glucose and lipid metabolism". Oncotarget (Review). 8 (1): 1845–1859. doi:10.18632/oncotarget.12157. PMC 5352102. PMID 27659520.
- Yamamoto H, Schoonjans K, Auwerx J (August 2007). "Sirtuin functions in health and disease". Molecular Endocrinology. 21 (8): 1745–55. doi:10.1210/me.2007-0079. PMID 17456799.
- Blander G, Guarente L (2004). "The Sir2 family of protein deacetylases". Annual Review of Biochemistry. 73 (1): 417–35. doi:10.1146/annurev.biochem.73.011303.073651. PMID 15189148.
- Wade N (2006-11-08). "The quest for a way around aging". Health & Science. International Herald Tribune. Retrieved 2008-11-30.
- "MIT researchers uncover new information about anti-aging gene". Massachusetts Institute of Technology, News Office. 2000-02-16. Retrieved 2008-11-30.
- Dryden SC, Nahhas FA, Nowak JE, Goustin AS, Tainsky MA (May 2003). "Role for human SIRT2 NAD-dependent deacetylase activity in control of mitotic exit in the cell cycle". Molecular and Cellular Biology. 23 (9): 3173–85. doi:10.1128/MCB.23.9.3173-3185.2003. PMC 153197. PMID 12697818.
- Zhao K, Chai X, Marmorstein R (March 2004). "Structure and substrate binding properties of cobB, a Sir2 homolog protein deacetylase from Escherichia coli". Journal of Molecular Biology. 337 (3): 731–41. doi:10.1016/j.jmb.2004.01.060. PMID 15019790.
- Schwer B, Verdin E (February 2008). "Conserved metabolic regulatory functions of sirtuins". Cell Metabolism. 7 (2): 104–12. doi:10.1016/j.cmet.2007.11.006. PMID 18249170.
- Choi JE, Mostoslavsky R (June 2014). "Sirtuins, metabolism, and DNA repair". Current Opinion in Genetics & Development. 26: 24–32. doi:10.1016/j.gde.2014.05.005. PMC 4254145. PMID 25005742.
- Aunan, JR; Watson, MM; Hagland, HR; Søreide, K (January 2016). "Molecular and biological hallmarks of ageing". The British Journal of Surgery (Review (in vitro)). 103 (2): e29-46. doi:10.1002/bjs.10053. PMID 26771470.
- López-Lluch, G; Navas, P (15 April 2016). "Calorie restriction as an intervention in ageing". The Journal of Physiology (Review). 594 (8): 2043–60. doi:10.1113/JP270543. PMC 4834802. PMID 26607973.
- Wang, Y; Liang, Y; Vanhoutte, PM (6 April 2011). "SIRT1 and AMPK in regulating mammalian senescence: a critical review and a working model". FEBS Letters (Review). 585 (7): 986–94. doi:10.1016/j.febslet.2010.11.047. hdl:10722/142456. PMID 21130086.
- Park, Seongjoon; Mori, Ryoichi; Shimokawa, Isao (8 May 2013). "Do sirtuins promote mammalian longevity?: A Critical review on its relevance to the longevity effect induced by calorie restriction". Molecules and Cells (Review). 35 (6): 474–480. doi:10.1007/s10059-013-0130-x. PMC 3887872. PMID 23661364.
- Smith DL, Jr; Nagy, TR; Allison, DB (May 2010). "Calorie restriction: what recent results suggest for the future of ageing research". European Journal of Clinical Investigation (Review). 40 (5): 440–50. doi:10.1111/j.1365-2362.2010.02276.x. PMC 3073505. PMID 20534066.
- Wyman AE, Atamas SP (March 2018). "Sirtuins and accelerated aging in scleroderma". Current Rheumatology Reports. 20 (4): 16. doi:10.1007/s11926-018-0724-6. PMC 5942182. PMID 29550994.
- Vazquez BN, Thackray JK, Serrano L (March 2017). "Sirtuins and DNA damage repair: SIRT7 comes to play". Nucleus. 8 (2): 107–115. doi:10.1080/19491034.2016.1264552. PMC 5403131. PMID 28406750.
- Uhl M, Csernok A, Aydin S, Kreienberg R, Wiesmüller L, Gatz SA (April 2010). "Role of SIRT1 in homologous recombination". DNA Repair. 9 (4): 383–93. doi:10.1016/j.dnarep.2009.12.020. PMID 20097625.
- Mostoslavsky R, Chua KF, Lombard DB, Pang WW, Fischer MR, Gellon L, Liu P, Mostoslavsky G, Franco S, Murphy MM, Mills KD, Patel P, Hsu JT, Hong AL, Ford E, Cheng HL, Kennedy C, Nunez N, Bronson R, Frendewey D, Auerbach W, Valenzuela D, Karow M, Hottiger MO, Hursting S, Barrett JC, Guarente L, Mulligan R, Demple B, Yancopoulos GD, Alt FW (January 2006). "Genomic instability and aging-like phenotype in the absence of mammalian SIRT6". Cell. 124 (2): 315–29. doi:10.1016/j.cell.2005.11.044. PMID 16439206.
- McCord RA, Michishita E, Hong T, Berber E, Boxer LD, Kusumoto R, Guan S, Shi X, Gozani O, Burlingame AL, Bohr VA, Chua KF (January 2009). "SIRT6 stabilizes DNA-dependent protein kinase at chromatin for DNA double-strand break repair". Aging. 1 (1): 109–21. doi:10.18632/aging.100011. PMC 2815768. PMID 20157594.
- Mao Z, Tian X, Van Meter M, Ke Z, Gorbunova V, Seluanov A (July 2012). "Sirtuin 6 (SIRT6) rescues the decline of homologous recombination repair during replicative senescence". Proceedings of the National Academy of Sciences of the United States of America. 109 (29): 11800–5. Bibcode:2012PNAS..10911800M. doi:10.1073/pnas.1200583109. PMC 3406824. PMID 22753495.
- Vazquez BN, Thackray JK, Simonet NG, Kane-Goldsmith N, Martinez-Redondo P, Nguyen T, Bunting S, Vaquero A, Tischfield JA, Serrano L (July 2016). "SIRT7 promotes genome integrity and modulates non-homologous end joining DNA repair". The EMBO Journal. 35 (14): 1488–503. doi:10.15252/embj.201593499. PMC 4884211. PMID 27225932.
- Avalos JL, Bever KM, Wolberger C (March 2005). "Mechanism of sirtuin inhibition by nicotinamide: altering the NAD(+) cosubstrate specificity of a Sir2 enzyme". Molecular Cell. 17 (6): 855–68. doi:10.1016/j.molcel.2005.02.022. PMID 15780941.
External links
- Sirtuins at the US National Library of Medicine Medical Subject Headings (MeSH)