Niobium pentaiodide
 | |
| Names | |
|---|---|
| Other names
Niobium(V) iodide | |
| Identifiers | |
| ECHA InfoCard | 100.034.006 |
| Properties | |
| Nb2I10 | |
| Molar mass | 1475 |
| Appearance | yellow solid |
| Density | 5.30 g/cm3 |
| Melting point | 543 °C (1,009 °F; 816 K) sublimes |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
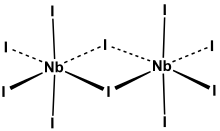
Niobium pentaiodide is the inorganic compound with the formula Nb2I10. It is a yellow, moisture-sensitive, diamagnetic solid. It forms upon heating metallic niobium with iodine.[1] Its structure has been verified by single crystal X-ray diffraction, which reveals an edge-shared bioctahedral structure, as observed for many pentahalides of Nb and Ta. The bonds to the bridging iodides are 0.3 Å longer than the terminal Nb-I bonds.[2]
References
- ↑ G. Brauer "Hydrogen, Deuterium, Water" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 2. p. 1315.
- ↑ Krebs, Bernt; Sinram, Diethard "Darstellung, Struktur und Eigenschaften einer neuen Modifikation von NbI5 (Preparation, structure and properties of a new modification of NbI5" Zeitschrift fǔr Naturforschung, Teil B: Anorganische Chemie, Organische Chemie 1980, volume 35b, pp. 12-16.
| HI | He | ||||||||||||||||
| LiI | BeI2 | BI3 | CI4 | NI3 | I2O4, I2O5, I4O9 |
IF, IF3, IF5, IF7 |
Ne | ||||||||||
| NaI | MgI2 | AlI3 | SiI4 | PI3, P2I4 |
S | ICl, ICl3 |
Ar | ||||||||||
| KI | CaI2 | Sc | TiI4 | VI3 | CrI3 | MnI2 | FeI2 | CoI2 | NiI2 | CuI | ZnI2 | Ga2I6 | GeI2, GeI4 |
AsI3 | Se | IBr | Kr |
| RbI | SrI2 | YI3 | ZrI4 | NbI5 | Mo | Tc | Ru | Rh | Pd | AgI | CdI2 | InI3 | SnI4, SnI2 |
SbI3 | TeI4 | I | Xe |
| CsI | BaI2 | HfI4 | TaI5 | W | Re | Os | Ir | Pt | AuI | Hg2I2, HgI2 |
TlI | PbI2 | BiI3 | Po | AtI | Rn | |
| Fr | Ra | Rf | Db | Sg | Bh | Hs | Mt | Ds | Rg | Cn | Nh | Fl | Mc | Lv | Ts | Og | |
| ↓ | |||||||||||||||||
| La | Ce | Pr | Nd | Pm | SmI2 | Eu | Gd | TbI3 | Dy | Ho | Er | Tm | Yb | Lu | |||
| Ac | ThI4 | Pa | UI3, UI4 |
Np | Pu | Am | Cm | Bk | Cf | Es | Fm | Md | No | Lr | |||
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.