Dithionic acid
 | |
 | |
| Names | |
|---|---|
| IUPAC name
dithionic acid [1] | |
| Other names
hypodisulfuric acid | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
PubChem CID |
|
| |
| |
| Properties | |
| H2S2O6 | |
| Molar mass | 162.14 g mol−1 |
| Conjugate base | Dithionate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
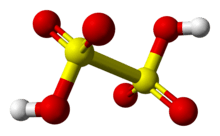
Dithionic acid, H2S2O6, is a chemical compound known only in solution.[2]
Salts
Dithionic acid is diprotic and salts called dithionates are known. No acid salts (i.e.: one proton lost) have been discovered. All dithionates are readily soluble in water.[2] They are mild oxidizing and mild reducing agents. The shape of the dithionate ion is like ethane, but two SO3 groups adopt an almost eclipsed conformation. The S—S bond length is about 2.15 Å; the S—O bonds are rather short with a bond length of 1.43 Å.
Synthesis
Dithionates can be made by oxidizing a sulfite (from the +4 to the +5 oxidation state), but on a larger scale they are made by oxidizing a cooled aqueous solution of sulfur dioxide with manganese dioxide:
- 2MnO2 + 3SO2 → MnS2O6 + MnSO4
The manganese dithionate solution formed can then be converted to dithionate salts of other metals by metathesis reactions:
- Ba2+(aq) + MnS2O6(aq) + MnSO4(aq) → BaSO4(s)↓ + BaS2O6·2H2O(aq)
Concentrated solutions of dithionic acid can subsequently be obtained treating a barium dithionate solution with sulfuric acid:
- BaS2O6(aq) + H2SO4(aq) → H2S2O6(aq) + BaSO4(s)↓
See also
Hypophosphoric acid the phosphorus equivalent.
References
- ↑ International Union of Pure and Applied Chemistry (2005). Nomenclature of Inorganic Chemistry (IUPAC Recommendations 2005). Cambridge (UK): RSC–IUPAC. ISBN 0-85404-438-8. p. 130. Electronic version.
- 1 2 Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9. pp. 715-716
www.chemindustry.com/chemicals/1022920.html - CASNo reference