Alpha secretase
Alpha secretases are a family of proteolytic enzymes that cleave amyloid precursor protein (APP) in its transmembrane region. Specifically, alpha secretases cleave within the fragment that gives rise to the Alzheimer's disease-associated peptide amyloid beta when APP is instead processed by beta secretase and gamma secretase. The alpha-secretase pathway is the predominant APP processing pathway. Thus, alpha-secretase cleavage precludes amyloid beta formation and is considered to be part of the non-amyloidogenic pathway in APP processing. Alpha secretases are members of the ADAM ('a disintegrin and metalloprotease domain') family, which are expressed on the surfaces of cells and anchored in the cell membrane. Several such proteins, notably ADAM10, have been identified as possessing alpha-secretase activity. Upon cleavage by alpha secretases, APP releases its extracellular domain - a fragment known as APPsα - into the extracellular environment in a process known as ectodomain shedding.[1]
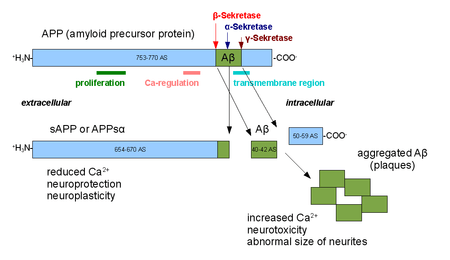
ADAM10 consists of two protein domains, a disintegrin domain and a prodomain; however, only the prodomain is required for APP processing.[2] Other ADAM proteins, ADAM17 (also called TACE, tumor necrosis factor-α converting enzyme),[3] ADAM9,[4] and ADAM19[5] have also been identified as alpha secretases; extracellular expression of mutant ADAM9 (also known as MDC9 or meltrin gamma) lacking the membrane anchor domain has been suggested as one of many possible means of Alzheimer's prevention and treatment exploiting the alpha secretase pathway.[6] Two distinct modalities of alpha-secretase activity have been observed in cells; constitutive activity occurs mainly at the cell surface [7] and is independent of regulatory mechanisms inside the cell, while regulated activity occurs mainly in the golgi and is dependent on the activity of protein kinase C. Alpha-secretase activity in the golgi is thought to compete directly with the beta-secretase pathway for APP substrates during membrane protein maturation.[8] Cell-surface cleavage by alpha secretase is very rapid after APP reaches the cell surface.[9]
The activity of alpha secretases has been implicated in the regulation of learning and memory formation. Release of the APPsα ectodomain has neurotrophic effects that counteract apoptotic signaling and promote synapse formation, processes that are upregulated when ADAM10 is overexpressed.[10] Alpha secretase activity has also been observed to be upregulated in response to the signaling peptide PACAP.[11]
Related alpha-secretases, including ADAM10, have also been implicated in similar maturation events for other transmembrane proteins such as MHC class I proteins. Recent evidence suggests that some such proteins are first processed to ectodomains by alpha secretases and subsequently cleaved by another Alzheimer's-associated protease complex, gamma secretase in its presenilin-complexed form.[12] The Notch pathway bears many similarities to APP processing and is also regulated in part by ADAM10.[13]
References
- ↑ Lammich, S; Kojro, E; Postina, R; Gilbert, S; Pfeiffer, R; Jasionowski, M; Haass, C; Fahrenholz, F (1999). "Constitutive and regulated alpha-secretase cleavage of Alzheimer's amyloid precursor protein by a disintegrin metalloprotease". Proc Natl Acad Sci USA. 96 (7): 3922–7. doi:10.1073/pnas.96.7.3922. PMC 22396. PMID 10097139.
- ↑ Fahrenholz, F; Gilbert, S; Kojro, E; Lammich, S; Postina, R (2000). "Alpha-secretase activity of the disintegrin metalloprotease ADAM 10. Influences of domain structure". Ann NY Acad Sci. 920: 215–22. doi:10.1111/j.1749-6632.2000.tb06925.x. PMID 11193153.
- ↑ Detlev Ganten, Aloys Greither : Molekularmedizinische Grundlagen von altersspezifischen Erkrankungen, 2004, Springer-Verlag, ISBN 3-540-00858-6
- ↑ Asai, M; Hattori, C; Szabo, B; Sasagawa, N; Maruyama, K; Tanuma, S; Ishiura, S (2003). "Putative function of ADAM9, ADAM10, and ADAM17 as APP alpha-secretase". Biochem Biophys Res Commun. 301 (1): 231–5. doi:10.1016/S0006-291X(02)02999-6. PMID 12535668.
- ↑ Tanabe, C; Hotoda, N; Sasagawa, N; Sehara-Fujisawa, A; Maruyama, K; Ishiura, S (2006). "ADAM19 is tightly associated with constitutive Alzheimer's disease APP alpha-secretase in A172 cells". Biochem Biophys Res Commun. 352 (1): 111–7. doi:10.1016/j.bbrc.2006.10.181. PMID 17112471.
- ↑ Hotoda, N; Koike, H; Sasagawa, N; Ishiura, S (2002). "A secreted form of human ADAM9 has an alpha-secretase activity for APP". Biochem Biophys Res Commun. 293 (2): 800–5. doi:10.1016/S0006-291X(02)00302-9. PMID 12054541.
- ↑ citation needed
- ↑ Skovronsky, DM; Moore, DB; Milla, ME; Doms, RW; Lee, VM (2000). "Protein kinase C-dependent alpha-secretase competes with beta-secretase for cleavage of amyloid-beta precursor protein in the trans-golgi network". J Biol Chem. 275 (4): 2568–75. doi:10.1074/jbc.275.4.2568. PMID 10644715.
- ↑ De Strooper, B; Annaert, W (2000). "Proteolytic processing and cell biological functions of the amyloid precursor protein". J Cell Sci. 113 (11): 1857–70. PMID 10806097.
- ↑ Bell, KF; Zheng, L; Fahrenholz, F; Cuello, AC (2006). "ADAM-10 over-expression increases cortical synaptogenesis". Neurobiol Aging. 29 (4): 554–65. doi:10.1016/j.neurobiolaging.2006.11.004. PMID 17187903.
- ↑ Kojro, E; Postina, R; Buro, C; Meiringer, C; Gehrig-Burger, K; Fahrenholz, F (2006). "The neuropeptide PACAP promotes the alpha-secretase pathway for processing the Alzheimer amyloid precursor protein". FASEB J. 20 (3): 512–4. doi:10.1096/fj.05-4812fje. PMID 16401644.
- ↑ Carey, BW; Kim, DY; Kovacs, DM (2007). "Presenilin/gamma-secretase and alpha-secretase-like peptidases cleave human MHC Class I proteins". Biochem J. 401 (1): 121–7. doi:10.1042/bj20060847. PMC 1698663. PMID 17150042.
- ↑ Hartmann, D; de Strooper, B; Serneels, L; Craessaerts, K; Herreman, A; Annaert, W; Umans, L; Lubke, T; Lena Illert, A; von Figura, K; Saftig, P (2002). "The disintegrin/metalloprotease ADAM 10 is essential for Notch signalling but not for alpha-secretase activity in fibroblasts". Hum Mol Genet. 11 (21): 2615–24. doi:10.1093/hmg/11.21.2615. PMID 12354787.