Organotantalum chemistry
Organotantalum chemistry is the chemistry of chemical compounds containing a carbon-to-tantalum chemical bond. A wide variety of compound have been reported, initially with cyclopentadienyl and CO ligands. Oxidation states vary from Ta(V) to Ta(-I).
Alkyl/aryl and alkylidene complexes
Pentamethyltantalum was reported by Richard Schrock in 1974.[1]
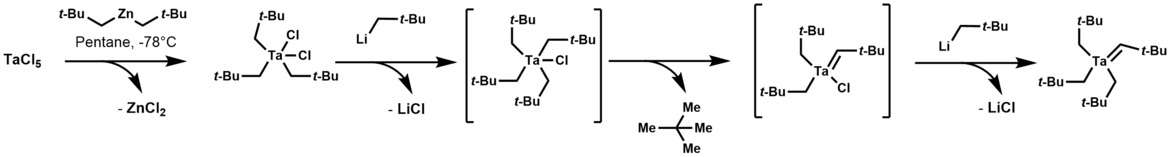
Tantalum alkylidene complexes were reported in 1979. Tantalum alkylidenes arise by treating trialkyltantalum dichloride with alkyl-lithium reagents. This classic conversion initially forms a thermally unstable tetraalkyl-monochloro-tantalum complex, which after α-hydrogen abstraction (a hydrogen on a carbon atom that has a bond to tantalum) and addition of another equivalent of alkyl-lithium, yields the alkylidene complex.[2]
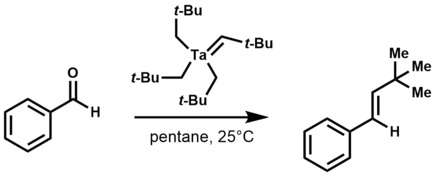
Tantalum alkylidene complexes are nucleophilic in contrast to Fischer-type carbene complexes, which are electrophilic at the alpha carbon.[2] They effect a number of reactions including: olefinations, olefin metathesis, hydroaminoalkylation of olefins, and conjugate allylation of enones.
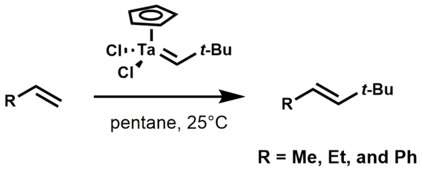
Ethylene, propylene, and styrene react readily with tantalum alkylidene complexes to yield olefin metathesis products.[3]
Cyclopentadienyl complexes
Some of the earliest organotantalum complexes to be prepared are the cyclopentadienyl derivatives. These arise from the salt metathesis reactions of sodium cyclopentadienide and tantalum pentachloride. More soluble and better developed are derivatives of pentamethylcyclopentadiene (Cp*H). Representative derivatives include Cp*TaCl4, Cp*2TaCl2, and Cp*2TaH3.[4]
Tantalum carbonyls and arenes
Treatment of tantalum pentachloride with hexamethylbenzene (C6Me6), aluminium and aluminium trichloride gives M(�η6-C6Me6)AlCl4]2.[5]
Reduction of TaCl5 under an atmosphere of CO gives the salts of [Ta(CO)6]−.[6] These same anions can be obtained by carbonylation of tantalum bis arene complex]]es.
Tantalum-alkyne complexes
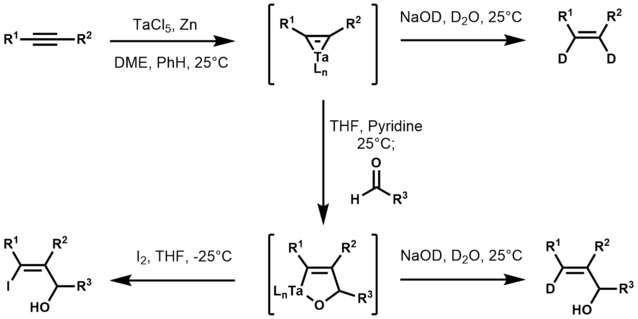
Although uncharacterized, tantalum-alkyne complexes were first claimed in the 1970s.[7] Some catalyze cyclotrimerizations.[8][9] The synthetic utility of tantalum-alkyne complexes was later expanded by K. Utimoto in 1989 when he used tantalum-alkyne complexes to synthesize trisubstituted allylic alcohols from acetylenes and aldehydes.[10] In this work, Utimoto first reduces tantalum(V) chloride with zinc to produce a low-valent tantalum species, which readily reacts with acetylenes to yield a tantalum-cyclopropene. Treatment of the tantalum-cyclopropene with THF, pyridine, and an aldehyde results in the formation of an oxatantala-cyclopentene, which upon aqueous work-up affords (E)-allylic alcohols exclusively. Utimoto also noted that treatment of the oxatantala-cyclopentene with iodine resulted in the corresponding iodo alcohol.
Tantalum-amido complexes
Organotantalum compounds are invoked as intermediates in C-alkylation of secondary amines with 1-alkenes using Ta(NMe2)5.[11] The chemistry developed by Maspero was later brought to fruition when Hartwig and Herzon reported the hydroaminoalkylation of olefins to form alkylamines:[12]
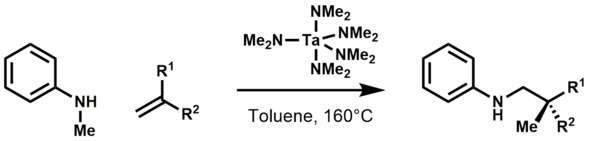
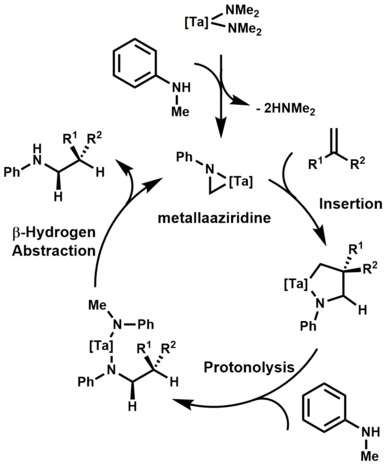
Mechanistically, the first step of the catalytic cycle is believed to be the C-H activation (via β-hydrogen abstraction) of the bisamide, which results in the formation of a metallaaziridine. Subsequent olefin insertion, protonolysis of the tantalum-carbon bond, and β-hydrogen abstraction affords the alkylamine product.[13] Notable advancements in this area were also made by Doye[14] and Schafer[15] when they showed tantalum catalyzed hydroaminoalkylations were exclusively branch selective and highly diastereoselective.
Transmetalation
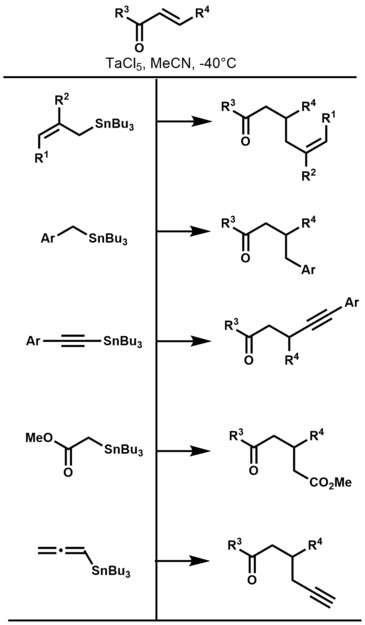
In 2002, Shibata and Baba reported the generation of organotantalum reagents via transmetalation of organotin compounds with tantalum(V) chloride and their subsequent conjugate addition to enones.[16] Of particular interest is the ability of certain organotantalum reagents to promote the conjugate allylation of enones. Though the direct allylation of carbonyl groups is prevalent throughout the literature, little has been reported on the conjugate allylation of enones.[17] Prior to Shibata and Baba's report, only three methods existed to selectively allylate enones, via: Hosomi Sakurai reaction (allylsilane and TiCl4),[18] allylbarium reagents,[19] and allylcopper reagents.[20] Transmetalation of allyltin, benzyltin, alkynyltin, α-stannyl esters, and allenyltin compounds with TaCl5 at cryogenic temperatures, efficiently resulted in the corresponding organotantalum compounds. The products formed via the conjugate addition of organotantalum compounds to enones are depicted below.
See also
- Compounds of carbon with other elements in the periodic table:
| Compounds of carbon with other elements in the periodic table | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
References
- ↑ Schrock, R (1974). "Pentamethyl Complexes of Niobium and Tantalum": 5288. doi:10.1021/ar50135a004.
- 1 2 Schrock, Richard R. (1979-03-01). "Alkylidene complexes of niobium and tantalum". Accounts of Chemical Research. 12 (3): 98–104. doi:10.1021/ar50135a004. ISSN 0001-4842.
- ↑ McLain, S. J.; Wood, C. D.; Schrock, R. R. (1977-05-01). "Multiple metal-carbon bonds. 6. The reaction of niobium and tantalum neopentylidene complexes with simple olefins: a route to metallocyclopentanes". Journal of the American Chemical Society. 99 (10): 3519–3520. doi:10.1021/ja00452a064. ISSN 0002-7863.
- ↑ Endy Y.-J. Min, John E. Bercaw (2014). "Bis(η5-Pentamethylcyclopentadienyl) Complexes of Niobium and Tantalum". Inorg. Synth. 36: 52–57. doi:10.1002/9781118744994.ch11.
- ↑ Pampaloni, G. (2010). "Aromatic hydrocarbons as ligands. Recent advances in the synthesis, the reactivity and the applications of bis(η6-arene) complexes". Coordination Chemistry Reviews. 254: 402–419. doi:10.1016/j.ccr.2009.05.014.
- ↑ J. E. Ellis, A. Davison, (1976). "Tris[Bis(2-Methoxyethyl)Ether]Potassium and Tetraphenylarsonium Hexacarbonylmetallates(1–) of Niobium and Tantalum". Inorg. Synth. 16: 68–73. doi:10.1002/9780470132470.ch21.
- ↑ Labinger, Jay A.; Schwartz, Jeffrey; Townsend, John M. (1974-06-01). "Iodo- and hydridotantalum(III) complexes of dialkylacetylenes". Journal of the American Chemical Society. 96 (12): 4009–4011. doi:10.1021/ja00819a047. ISSN 0002-7863.
- ↑ Cotton, F. Albert; Hall, William T. (1979-08-01). "Reactions of tantalum(III) with alkynes and nitriles". Journal of the American Chemical Society. 101 (17): 5094–5095. doi:10.1021/ja00511a064. ISSN 0002-7863.
- ↑ Bruck, M. A.; Copenhaver, A. S.; Wigley, D. E. (1987-10-01). "Alkyne cyclizations at reduced tantalum centers: synthesis and molecular structure of (.eta.6-C6Me6)Ta(O-2,6-i-Pr2C6H3)2Cl". Journal of the American Chemical Society. 109 (21): 6525–6527. doi:10.1021/ja00255a056. ISSN 0002-7863.
- ↑ Takai, Kazuhiko; Kataoka, Y.; Utimoto, K. (1990-03-01). "Tantalum-alkyne complexes as synthetic intermediates. Stereoselective preparation of trisubstituted allylic alcohols from acetylenes and aldehydes". The Journal of Organic Chemistry. 55 (6): 1707–1708. doi:10.1021/jo00293a008. ISSN 0022-3263.
- ↑ Clerici, Mario G.; Maspero, Federico (1980-01-01). "Catalytic C-Alkylation of Secondary Amines with Alkenes". Synthesis. 1980 (04): 305–306. doi:10.1055/s-1980-29002. ISSN 0039-7881.
- ↑ Herzon, Seth B.; Hartwig, John F. (2007-05-01). "Direct, Catalytic Hydroaminoalkylation of Unactivated Olefins with N-Alkyl Arylamines". Journal of the American Chemical Society. 129 (21): 6690–6691. doi:10.1021/ja0718366. ISSN 0002-7863. PMC 2590937. PMID 17474747.
- ↑ Eisenberger, Patrick; Ayinla, Rashidat O.; Lauzon, Jean Michel P.; Schafer, Laurel L. (2009-10-19). "Tantalum–Amidate Complexes for the Hydroaminoalkylation of Secondary Amines: Enhanced Substrate Scope and Enantioselective Chiral Amine Synthesis". Angewandte Chemie International Edition. 48 (44): 8361–8365. doi:10.1002/anie.200903656. ISSN 1521-3773.
- ↑ Dörfler, Jaika; Doye, Sven (2014-05-01). "A Commercially Available Tantalum Catalyst for the Highly Regioselective Intermolecular Hydroaminoalkylation of Styrenes". European Journal of Organic Chemistry. 2014 (13): 2790–2797. doi:10.1002/ejoc.201400082. ISSN 1099-0690.
- ↑ Payne, Philippa R.; Garcia, Pierre; Eisenberger, Patrick; Yim, Jacky C.-H.; Schafer, Laurel L. (2013-05-03). "Tantalum Catalyzed Hydroaminoalkylation for the Synthesis of α- and β-Substituted N-Heterocycles". Organic Letters. 15 (9): 2182–2185. doi:10.1021/ol400729v. ISSN 1523-7060.
- ↑ Shibata, Ikuya; Kano, Takeyoshi; Kanazawa, Nobuaki; Fukuoka, Shoji; Baba, Akio (2002-04-15). "Generation of Organotantalum Reagents and Conjugate Addition to Enones". Angewandte Chemie. 114 (8): 1447–1450. doi:10.1002/1521-3757(20020415)114:83.0.CO;2-B. ISSN 1521-3757.
- ↑ Yamamoto, Yoshinori.; Asao, Naoki. (1993-09-01). "Selective reactions using allylic metals". Chemical Reviews. 93 (6): 2207–2293. doi:10.1021/cr00022a010. ISSN 0009-2665.
- ↑ Hosomi, Akira; Sakurai, Hideki (1977-03-01). "Chemistry of organosilicon compounds. 99. Conjugate addition of allylsilanes to alpha,beta-enones. A New method of stereoselective introduction of the angular allyl group in fused cyclic alpha,beta-enones". Journal of the American Chemical Society. 99 (5): 1673–1675. doi:10.1021/ja00447a080. ISSN 0002-7863.
- ↑ Yanagisawa, Akira; Habaue, Shigeki; Yasue, Katsutaka; Yamamoto, Hisashi (1994-07-01). "Allylbarium Reagents: Unprecedented Regio- and Stereoselective Allylation Reactions of Carbonyl Compounds". Journal of the American Chemical Society. 116 (14): 6130–6141. doi:10.1021/ja00093a010. ISSN 0002-7863.
- ↑ Lipshutz, Bruce H.; Ellsworth, Edmund L.; Dimock, Stuart H.; Smith, Robin A. J. (1990-05-01). "New methodology for conjugate additions of allylic ligands to .alpha.,.beta.-unsaturated ketones: synthetic and spectroscopic studies". Journal of the American Chemical Society. 112 (11): 4404–4410. doi:10.1021/ja00167a043. ISSN 0002-7863.