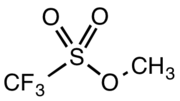
Methyl trifluoromethanesulfonate
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Methyl trifluoromethanesulfonate | |
| Other names
Trifluoromethanesulfonic acid, methyl ester Triflic acid, methyl ester, methyl triflate | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.005.793 |
PubChem CID |
|
| |
| |
| Properties | |
| C2H3F3O3S | |
| Molar mass | 164.10 g·mol−1 |
| Appearance | Colourless Liquid |
| Density | 1.496 g/mL |
| Melting point | −64 °C (−83 °F; 209 K) |
| Boiling point | 100 °C (212 °F; 373 K) |
| Hydrolyzes | |
| Hazards | |
| Main hazards | Corrosive |
| R-phrases (outdated) | R10-R34 |
| S-phrases (outdated) | S26-S36/37/39-S45 |
| Flash point | 38 °C (100 °F; 311 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Methyl trifluoromethanesulfonate, also commonly called methyl triflate and abbreviated MeOTf, is the organic compound with the formula CF3SO2OCH3. It is a colourless liquid which finds use in organic chemistry as a powerful methylating agent.[1] The compound is closely related to methyl fluorosulfonate (FSO2OCH3).
Synthesis
Methyl triflate is commercially available, however it may also be prepared in the laboratory by treating dimethyl sulfate with triflic acid.[2]
- CF3SO2OH + (CH3O)2SO2 → CF3SO2OCH3 + CH3OSO2OH
Reactivity
The compound hydrolyzes violently upon contact with water:
- CF3SO2OCH3 + H2O → CF3SO2OH + CH3OH
Methylation
One ranking of methylating agents is (CH3)3O+ > CF3SO2OCH3 ≈ FSO2OCH3 > (CH3)2SO4 > CH3I.[2] It will alkylate many functional groups that are only weakly basic such as aldehydes, amides, and nitriles. It does not methylate benzene or the bulky 2,6-di-tert-butylpyridine.[1] Its ability to methylate N-heterocycles is exploited in certain deprotection schemes.[3]
See also
References
- 1 2 Roger W. Alder; Justin G. E. Phillips; Lijun Huang; Xuefei Huang (2005). "Methyltrifluoromethanesulfonate". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rm266m.pub2.
- 1 2 Stang, Peter J.; Hanack, Michael; Subramanian, L. R. (1982). "Perfluoroalkanesulfonic Esters: Methods of Preparation and Applications in Organic Chemistry". Synthesis. 1982 (02): 85–126. doi:10.1055/s-1982-29711. ISSN 0039-7881.
- ↑ Albert I. Meyers; Mark E. Flanagan (1998). "2,2′-Dimethoxy-6-formylbiphenyl". Organic Syntheses. ; Collective Volume, 9, p. 258