Amide
In organic chemistry, an amide, also known as an organic amide or a carboxamide, is a compound with the general formula RC(=O)NR′R″, where R, R′, and R″ represent organic groups or hydrogen atoms.[1][2] It can be viewed as a derivative of a carboxylic acid RC(=O)OH with the hydroxyl group –OH replaced by an amine group –NR′R″; or, equivalently, an acyl (alkanoyl) group RC(=O)– joined to an amine group.

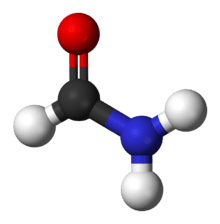

Common examples of amides are acetamide H3C–CONH2, benzamide C6H5–CONH2, and dimethylformamide HCON(–CH3)2.
Amides are qualified as primary, secondary, and tertiary according to whether the amine subgroup has the form –NH2, –NHR, or –NRR′, where R and R′ are groups other than hydrogen.
The core –C(=O)N= of amides is called the amide group (specifically, carboxamide group).
Amides are pervasive in nature and technology. Proteins and important plastics like Nylons, Aramid, Twaron, and Kevlar are polymers whose units are connected by amide groups (polyamides); these linkages are easily formed, confer structural rigidity, and resist hydrolysis. (In biochemistry, the amide group is called a peptide bond when it is part of the main chain of a protein, and isopeptide bond when it occurs in a side chain, such as in the amino acids asparagine and glutamine.) Amides include many other important biological compounds, as well as many drugs like paracetamol, penicillin and LSD.[3] Low molecular weight amides, such as dimethylformamide, are common solvents.
Nomenclature
In the usual nomenclature, one adds the term "amide" to the stem of the parent acid's name. For instance, the amide derived from acetic acid is named acetamide (CH3CONH2). IUPAC recommends ethanamide, but this and related formal names are rarely encountered. When the amide is derived from a primary or secondary amine, the substituents on nitrogen are indicated first in the name. Thus, the amide formed from dimethylamine and acetic acid is N,N-dimethylacetamide (CH3CONMe2, where Me = CH3). Usually even this name is simplified to dimethylacetamide. Cyclic amides are called lactams; they are necessarily secondary or tertiary amides.[4]
Properties
Bonding

The lone pair of electrons on the nitrogen atom is delocalized into the carbonyl group, thus forming a partial double bond between nitrogen and carbon. In fact the O, C and N atoms have molecular orbitals occupied by delocalized electrons, forming a conjugated system. Consequently, the three bonds of the nitrogen in amides is not pyramidal (as in the amines) but planar.
The structure of an amide can be described also as a resonance between two alternative structures:
It is estimated that for acetamide, structure A makes a 62% contribution to the structure, while structure B makes a 28% contribution. (These figures do not sum to 100% because there are additional less-important resonance forms that are not depicted above). There is also a hydrogen bond present between the active groups hydrogen and nitrogen atoms.[8] Resonance is largely prevented in the very strained quinuclidone.
Basicity
Compared to amines, amides are very weak bases. While the conjugate acid of an amine has a pKa of about 9.5, the conjugate acid of an amide has a pKa around −0.5. Therefore, amides don't have as clearly noticeable acid–base properties in water. This relative lack of basicity is explained by the withdrawing of electrons from the amine by the carbonyl. On the other hand, amides are much stronger bases than carboxylic acids, esters, aldehydes, and ketones (their conjugate acids' pKas are between −6 and −10).
The proton of a primary or secondary amide does not dissociate readily under normal conditions; its pKa is usually well above 15. Conversely, under extremely acidic conditions, the carbonyl oxygen can become protonated with a pKa of roughly −1. It is not only because of the positive charge on the nitrogen, but also because of the negative charge on the oxygen gained through resonance.
Hydrogen bonding and solubility
Because of the greater electronegativity of oxygen, the carbonyl (C=O) is a stronger dipole than the N–C dipole. The presence of a C=O dipole and, to a lesser extent a N–C dipole, allows amides to act as H-bond acceptors. In primary and secondary amides, the presence of N–H dipoles allows amides to function as H-bond donors as well. Thus amides can participate in hydrogen bonding with water and other protic solvents; the oxygen atom can accept hydrogen bonds from water and the N–H hydrogen atoms can donate H-bonds. As a result of interactions such as these, the water solubility of amides is greater than that of corresponding hydrocarbons. These hydrogen bonds are also have an important role in the secondary structure of proteins.
The solubilities of amides and esters are roughly comparable. Typically amides are less soluble than comparable amines and carboxylic acids since these compounds can both donate and accept hydrogen bonds. Tertiary amides, with the important exception of N,N-dimethylformamide, exhibit low solubility in water.
Characterization
The presence of the amide group –C(=O)N– is generally easily established, at least in small molecules. It can be distinguished from nitro and cyano groups in IR spectra. Amides exhibit a moderately intense νCO band near 1650 cm−1. By 1H NMR spectroscopy, CONHR signals occur at low fields. In X-ray crystallography, the C(=O)N center together with the three immediately adjacent atoms characteristically define a plane.
Reactions
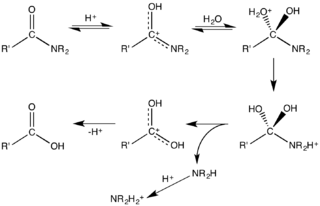 Mechanism for acid-mediated hydrolysis of an amide.[9]
Mechanism for acid-mediated hydrolysis of an amide.[9]
Amides undergo many chemical reactions, although they are less reactive than esters. Amides hydrolyse in hot alkali as well as in strong acidic conditions. Acidic conditions yield the carboxylic acid and the ammonium ion while basic hydrolysis yield the carboxylate ion and ammonia. The protonation of the initially generated amine under acidic conditions and the deprotonation of the initially generated carboxylic acid under basic conditions render these processes non-catalytic and irreversible. Amides are also versatile precursors to many other functional groups. Electrophiles attack the carbonyl oxygen. This step often precedes hydrolysis, which is catalyzed by both Brønsted acids and Lewis acids. Enzymes, e.g. peptidases and artificial catalysts, are known to accelerate the hydrolysis reactions.
| Reaction name | Product | Comment |
|---|---|---|
| Dehydration | Nitrile | Reagent: phosphorus pentoxide; benzenesulfonyl chloride; TFAA/py[10] |
| Hofmann rearrangement | Amine with one fewer carbon atom | Reagents: bromine and sodium hydroxide |
| Amide reduction | Amine | Reagent: lithium aluminium hydride followed by hydrolysis |
| Vilsmeier–Haack reaction | Aldehyde (via imine) | POCl3, aromatic substrate, formamide |
| Bischler–Napieralski reaction | Cyclic imine | POCl3, SOCl2, etc. |
Synthesis
Many methods exist in amide synthesis.[11] On paper, the simplest method for making amides is by coupling a carboxylic acid with an amine. In general this reaction is thermodynamically favorable, however it suffers from a high activation energy, largely due to the amine first deprotonating the carboxylic acid, which reduces its reactivity. As such the direct reaction often requires high temperatures.
- RCO2H + R′R″NH ⇌ RCO−
2 + R′R″NH+
2 ⇌ RC(O)NR′R″ + H2O
Many methods are known for driving the equilibrium to the right. For the most part these reactions involve "activating" the carboxylic acid by first converting it to a better electrophile; such as esters, acid chlorides (Schotten-Baumann reaction) or anhydrides (Lumière–Barbier method). Conventional methods in peptide synthesis use coupling agents such as HATU, HOBt, or PyBOP.[12] In recent years there has also been a surge in the development of Boron reagents for amide bond formation, including catalytic use of 2-IodoPhenylBoronic acid[13] or MIBA,[14] and Tris(2,2,2-trifluoroethyl) borate.[15][16]
| Reaction name | Substrate | Details |
|---|---|---|
| Nucleophilic acyl substitution | acyl chloride or acid anhydride | Reagent: ammonia or amines |
| Beckmann rearrangement | Cyclic ketone | Reagent: hydroxylamine and acid |
| Schmidt reaction | Ketones | Reagent: hydrazoic acid |
| Nitrile hydrolysis[17] | Nitrile | Reagent: water; acid catalyst |
| Willgerodt–Kindler reaction | Aryl alkyl ketones | Sulfur and morpholine |
| Passerini reaction | Carboxylic acid, ketone or aldehyde | |
| Ugi reaction | Isocyanide, carboxylic acid, ketone, primary amine | |
| Bodroux reaction[18][19] | Carboxylic acid, Grignard reagent with an aniline derivative ArNHR′ | 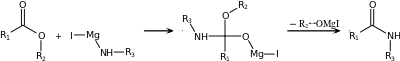 |
| Chapman rearrangement[20][21] | Aryl imino ether | For N,N-diaryl amides. The reaction mechanism is based on a nucleophilic aromatic substitution.[22] 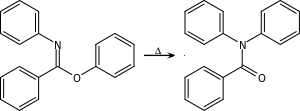 Chapman Rearrangement |
| Leuckart amide synthesis[23] | Isocyanate | Reaction of arene with isocyanate catalysed by aluminium trichloride, formation of aromatic amide. |
| Ritter reaction[24] | Alkenes, alcohols, or other carbonium ion sources | Secondary amides via an addition reaction between a nitrile and a carbonium ion in the presence of concentrated acids. |
| Photolytic addition of formamide to olefins[25] | Terminal alkenes | A free radical homologation reaction between a terminal alkene and formamide. |
| Ester aminolysis[26][27][28] | Esters | Base catalyzed reaction of esters with various amines to form alcohols and amides. |
Other methods
Dehydrogenative acylation of amines is catalyzed by organoruthenium complexes:[29]
The reaction proceed by one dehydrogenation of the alcohol to the aldehyde followed by formation of a hemiaminal, which undergoes a second dehydrogenation to the amide. Elimination of water in the hemiaminal to the imine is not observed.
Transamidation is typically very slow, but it is accelerated with Lewis acid[30] and organometallic catalysts:[31]
- RC(O)NR'2 + HNR"2 → RC(O)NR"2 + HNR'2
Primary amides (RC(O)NH2) are more amenable to this reaction.
See also
- Amidogen
- Amino radical
- Amidicity
- Metal amides
References
- IUPAC, Compendium of Chemical Terminology, 2nd ed. (the "Gold Book") (1997). Online corrected version: (2006–) "amides". doi:10.1351/goldbook.A00266
- "Chapter 21: Amides and Imides". Nomenclature of Organic Compounds. Advances in Chemistry. Volume 126. Washington, American Chemical Society. 1974. pp. 166–173. doi:10.1021/ba-1974-0126.ch021. ISBN 9780841201910.
- Boonen, Jente; Bronselaer, Antoon; Nielandt, Joachim; Veryser, Lieselotte; De Tré, Guy; De Spiegeleer, Bart (2012). "Alkamid database: Chemistry, occurrence and functionality of plant N-alkylamides" (PDF). Journal of Ethnopharmacology. 142 (3): 563–90. doi:10.1016/j.jep.2012.05.038. hdl:1854/LU-2133714. PMID 22659196.
- Organic Chemistry IUPAC Nomenclature. Rules C-821. Amides http://www.acdlabs.com/iupac/nomenclature/79/r79_540.htm Archived 21 January 2011 at the Wayback Machine
- "Amide definition and meaning - Collins English Dictionary". www.collinsdictionary.com. Retrieved 15 April 2018.
- "amide". The American Heritage Dictionary of the English Language (5th ed.). Boston: Houghton Mifflin Harcourt.
- "amide - Definition of amide in English by Oxford Dictionaries". Oxford Dictionaries - English. Retrieved 15 April 2018.
- Kemnitz, Carl R.; Loewen, Mark J. (2007). ""Amide Resonance" Correlates with a Breadth of C−N Rotation Barriers". Journal of the American Chemical Society. 129 (9): 2521–8. doi:10.1021/ja0663024. PMID 17295481.
- Smith, Michael B.; March, Jerry (2007), Advanced Organic Chemistry: Reactions, Mechanisms, and Structure (6th ed.), New York: Wiley-Interscience, ISBN 978-0-471-72091-1
- U.S. Patent 5,935,953
- Montalbetti, Christian A. G. N.; Falque, Virginie (14 November 2005). "Amide bond formation and peptide coupling". Tetrahedron. 61 (46): 10827–10852. doi:10.1016/j.tet.2005.08.031.
- Valeur, Eric; Bradley, Mark (2009). "Amide bond formation: beyond the myth of coupling reagents". Chem. Soc. Rev. 38 (2): 606–631. doi:10.1039/B701677H. PMID 19169468.
- "Greener Methods: Catalytic Amide Bond Formation". Retrieved 22 September 2016.
- "MIBA 96% | Sigma-Aldrich". www.sigmaaldrich.com. Retrieved 22 September 2016.
- "Tris(2,2,2-trifluoroethyl) borate 97% | Sigma-Aldrich". www.sigmaaldrich.com. Retrieved 22 September 2016.
- Sabatini, Marco T.; Boulton, Lee T.; Sheppard, Tom D. (1 September 2017). "Borate esters: Simple catalysts for the sustainable synthesis of complex amides". Science Advances. 3 (9): e1701028. Bibcode:2017SciA....3E1028S. doi:10.1126/sciadv.1701028. PMC 5609808. PMID 28948222.
- Wenner, Wilhelm (1952). "Phenylacetamide". Organic Syntheses. 32: 92. doi:10.15227/orgsyn.032.0092.
- Bodroux F. (1905). Bull. Soc. Chim. France. 33: 831.CS1 maint: untitled periodical (link)
- "Bodroux reaction". Institute of Chemistry, Skopje, Macedonia.
- Schulenberg, J. W.; Archer, S. (1965). The Chapman Rearrangement. Org. React. 14. pp. 1–51. doi:10.1002/0471264180.or014.01. ISBN 978-0471264187.
- Chapman, Arthur William (1925). "CCLXIX.—Imino-aryl ethers. Part III. The molecular rearrangement of N-phenylbenziminophenyl ether". Journal of the Chemical Society, Transactions. 127: 1992–1998. doi:10.1039/CT9252701992.
- March, Jerry (1966). Advanced organic Chemistry, Reactions, mechanisms and structure (3rd ed.). ISBN 978-0-471-85472-2.
- Leuckart, R. (1885). "Ueber einige Reaktionen der aromatischen Cyanate". Berichte der deutschen chemischen Gesellschaft. 18: 873–877. doi:10.1002/cber.188501801182.
- Adams, Rodger; Krimen, L.I.; Cota, Donald J. (1969). Organic Reaction Volume 17. London: John Wiley & Sons, Inc. pp. 213–326. doi:10.1002/0471264180. ISBN 9780471196150.
- Monson, Richard (1971). Advanced Organic Synthesis: Methods and Techniques (PDF). Newyork: Academic Press. p. 141. ISBN 978-0124336803.
- Corson, B. B.; Scott, R. W.; Vose, C. E. (1941). "Cyanoacetamide". Organic Syntheses. 1: 179. doi:10.15227/orgsyn.009.0036.
- Jacobs, W. A. (1941). "Chloroacetamide". Organic Syntheses. 1: 153. doi:10.15227/orgsyn.007.0016.
- Kleinberg, J.; Audrieth, L. F. (1955). "Lactamide". Organic Syntheses. 3: 516. doi:10.15227/orgsyn.021.0071.
- Gunanathan, C.; Ben-David, Y.; Milstein, D. (2007). "Direct Synthesis of Amides from Alcohols and Amines with Liberation of H2". Science. 317 (5839): 790–2. Bibcode:2007Sci...317..790G. doi:10.1126/science.1145295. PMID 17690291.
- T. A. Dineen, M. A. Zajac, A. G. Myers (2006). "Efficient Transamidation of Primary Carboxamides by in situ Activation with N,N-Dialkylformamide Dimethyl Acetals". J. Am. Chem. Soc. 128 (50): 16406–16409. doi:10.1021/ja066728i. PMID 17165798.CS1 maint: uses authors parameter (link)
- Emma L. Baker, Michael M. Yamano, Yujing Zhou, Sarah M. Anthony, Neil K. Garg (2016). "A two-step approach to achieve secondary amide transamidation enabled by nickel catalysis". Nature Communications. 7: 11554. Bibcode:2016NatCo...711554B. doi:10.1038/ncomms11554. PMC 4876455. PMID 27199089.CS1 maint: uses authors parameter (link)
External links
| Wikiquote has quotations related to: Amide |
- Amide synthesis (coupling reaction) – Synthetic protocols from organic-reaction.com
- IUPAC Compendium of Chemical Terminology

