Hypoiodous acid
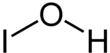
Hypoiodous acid is the inorganic compound with the chemical formula HOI. It forms when an aqueous solution of iodine is treated with mercuric or silver salts. It rapidly decomposes by disproportionation:[1]
- 5 HOI → HIO3 + 2I2 + 2H2O
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Hypoiodous acid | |||
| Other names
Hypoiodous acid | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
PubChem CID |
|||
| UNII | |||
CompTox Dashboard (EPA) |
|||
| |||
| |||
| Properties | |||
| HOI | |||
| Molar mass | 143.89 g/mol | ||
| Acidity (pKa) | 10.5 (in water, estimate) | ||
| Conjugate base | Hypoiodite | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Hypoiodites of alkali and alkaline earth metals can be made in cold dilute solutions if iodine is added to their respective hydroxides. Hypoiodous acid is a weak acid with a pKa of about 11. The conjugate base is hypoiodite (IO−). Salts of this anion can be prepared by treating I2 with alkali hydroxides. They rapidly disproportionate to form iodides and iodates.[1]
References
- Holleman, A.F. (2001). Inorganic chemistry (1st English ed., [edited] by Nils Wiberg. ed.). San Diego, Calif. : Berlin: Academic Press, W. de Gruyter. ISBN 0-12-352651-5.
This article is issued from Wikipedia. The text is licensed under Creative Commons - Attribution - Sharealike. Additional terms may apply for the media files.