Titanium(III) iodide
 | |
| Identifiers | |
|---|---|
| Properties | |
| I3Ti | |
| Molar mass | 428.58 g·mol−1 |
| Appearance | black-violet solid |
| Density | 4.96 g·cm−3[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Titanium(III) iodide is an inorganic compound with the formula TiI3. It is a dark violet solid that is insoluble in solvents, except upon decomposition.
Preparation and structure
Titanium(III) iodide can be prepared by reaction of titanium with iodine:[2]
It can also be obtained by reduction of TiI4, e.g., with aluminium .[3]
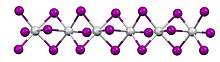
In terms of its structure, the compound exists as a polymer of face-sharing octahedra. Above 323 K, the Ti---Ti spacing are equal, but below that temperature, the material undergoes a phase transition. In the low temperature phase, the Ti---Ti contacts are alternating short and long. The low temperature structure is similar to that of molybdenum tribromide.[1]
References
- 1 2 Joachim Angelkort, Andreas Schoenleber, Sander van Smaalen: Low- and high-temperature crystal structures of. In: Journal of Solid State Chemistry. 182, 2009, S. 525–531, doi:10.1016/j.jssc.2008.11.028.
- ↑ F. Hein, S. Herzog "Molybdenum(III) Bromide" in Handbook of Preparative Inorganic Chemistry, 2nd Ed. Edited by G. Brauer, Academic Press, 1963, NY. Vol. 1. p. 1407.
- ↑ Catherine E. Housecroft, A. G. Sharpe (2005) (in German), [, p. 601, at Google Books Inorganic Chemistry], Pearson Education, pp. 601, ISBN 0-13039913-2, , p. 601, at Google Books
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.