JWH-176
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| Chemical and physical data | |
| Formula | C25H24 |
| Molar mass | 324.457 g/mol |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
JWH-176 is an analgesic drug which acts as a cannabinoid receptor agonist. Its binding affinity at the CB1 receptor is only 26.0nM, making it more potent than THC itself,[1] however JWH-176 is particularly notable in that it is a hydrocarbon containing no heteroatoms. This demonstrates that reasonably high-affinity cannabinoid binding and agonist effects can be produced by compounds with no hydrogen bonding capacity at all, relying merely on Van der Waals interactions to bind to the receptor.[2] It was discovered by, and named after, Dr. John W. Huffman.
Stereochemistry
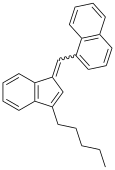
JWH-171
JWH-176 is the (E) stereoisomer of 1-([3-pentylinden-1-ylidine]methyl)naphthalene, whereas JWH-171 is the mixture of the (E) and (Z) isomers.[3]
See also
References
- ↑ Huffman JW, Padgett LW. Recent Developments in the Medicinal Chemistry of Cannabimimetic Indoles, Pyrroles and Indenes. Current Medicinal Chemistry, 2005; 12: 1395-1411.
- ↑ Roger Pertwee. Cannabinoids. Handbook of Experimental Pharmacology Volume 168, p 269. Springer. ISBN 3-540-22565-X
- ↑ Rahman, Atta-ur; Iqbal Choudhary, M; Reitz, Allen B (2010-12-10). Frontiers in Medicinal Chemistry , Volume (4). p. 681. ISBN 978-1608052073.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.