FDU-PB-22
 | |
| Legal status | |
|---|---|
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| Chemical and physical data | |
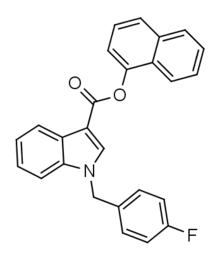
| Formula | C26H18FNO2 |
| Molar mass | 395.42 g/mol |
| 3D model (JSmol) | |
| |
| |
FDU-PB-22 is an derivative of JWH-018 that is presumed to be a potent agonist of the CB1 receptor and has been sold online as a designer drug.[1][2]
Pharmacology
FDU-PB-22 acts as a full agonist with a binding affinity of 1.19nM at CB1 and 2.43nM at CB2 cannabinoid receptors.[3]
Legal status
FDU-PB-22 is a controlled substance in Germany and is banned in Japan and Sweden.[4]
See also
References
- ↑ "FDU-PB-22". Southern Association of Forensic Scientists. Retrieved 23 July 2015.
- ↑ Uchiyama, Nahoko; Shimokawa, Yoshihiko; Kikura-Hanajiri, Ruri; Demizu, Yosuke; Goda, Yukihiro; Hakamatsuka, Takashi (1 July 2015). "A synthetic cannabinoid FDU-NNEI, two 2H-indazole isomers of synthetic cannabinoids AB-CHMINACA and NNEI indazole analog (MN-18), a phenethylamine derivative N–OH-EDMA, and a cathinone derivative dimethoxy-α-PHP, newly identified in illegal products". Forensic Toxicology. 33 (2): 244–259. doi:10.1007/s11419-015-0268-7. ISSN 1860-8965. PMC 4525202. PMID 26257833.
- ↑ Hess, Cornelius; Schoeder, ClaraT.; Pillaiyar, Thanigaimalai; Madea, Burkhard; Müller, ChristaE. (1 July 2016). "Pharmacological evaluation of synthetic cannabinoids identified as constituents of spice". Forensic Toxicology. 34 (2): 329–343. doi:10.1007/s11419-016-0320-2. ISSN 1860-8965. PMC 4929166. PMID 27429655.
- ↑ "Cannabinoider föreslås bli klassificerade som hälsofarlig vara". Folkhälsomyndigheten. 28 May 2014. Retrieved 23 July 2015.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.