Indium phosphide
 | |
 | |
| Names | |
|---|---|
| Other names
Indium(III) phosphide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| ECHA InfoCard | 100.040.856 |
PubChem CID |
|
| |
| |
| Properties | |
| InP | |
| Molar mass | 145.792 g/mol |
| Appearance | black cubic crystals |
| Density | 4.81 g/cm3, solid |
| Melting point | 1,062 °C (1,944 °F; 1,335 K) |
| Solubility | slightly soluble in acids[1] |
| Band gap | 1.344 eV (300 K; direct) |
| Electron mobility | 5400 cm2/(V·s) (300 K) |
| Thermal conductivity | 0.68 W/(cm·K) (300 K) |
Refractive index (nD) |
3.1 (infrared); 3.55 (632.8 nm)[2] |
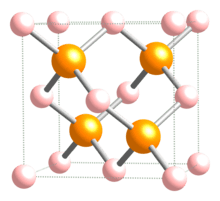
| Structure | |
| Zinc blende | |
| Tetrahedral | |
| Thermochemistry | |
Heat capacity (C) |
45.4 J/(mol·K)[4] |
Std molar entropy (S |
59.8 J/(mol·K) |
Std enthalpy of formation (ΔfH |
-88.7 kJ/mol |
| Hazards | |
| Main hazards | Toxic, hydrolysis to phosphine |
| Safety data sheet | External MSDS |
| Related compounds | |
Other anions |
Indium nitride Indium arsenide Indium antimonide |
Other cations |
Aluminium phosphide Gallium phosphide |
Related compounds |
Indium gallium phosphide Aluminium gallium indium phosphide Gallium indium arsenide antimonide phosphide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Indium phosphide (InP) is a binary semiconductor composed of indium and phosphorus. It has a face-centered cubic ("zincblende") crystal structure, identical to that of GaAs and most of the III-V semiconductors.
Manufacturing
.jpg)
Indium phosphide can be prepared from the reaction of white phosphorus and indium iodide at 400 °C.,[5] also by direct combination of the purified elements at high temperature and pressure, or by thermal decomposition of a mixture of a trialkyl indium compound and phosphine.[6]
Uses
InP is used in high-power and high-frequency electronics because of its superior electron velocity with respect to the more common semiconductors silicon and gallium arsenide.
It was used with indium gallium arsenide to make a record breaking pseudomorphic heterojunction bipolar transistor that could operate at 604 GHz.[7]
It also has a direct bandgap, making it useful for optoelectronics devices like laser diodes. The company Infinera uses indium phosphide as its major technological material for manufacturing photonic integrated circuits for the optical telecommunications industry, to enable wavelength-division multiplexing [8] applications.
InP is also used as a substrate for epitaxial indium gallium arsenide based opto-electronic devices.
Chemistry
Indium phosphide also has one of the longest-lived optical phonons of any compound with the zincblende crystal structure.[9]
References
- ↑ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 4–61, ISBN 0-8493-0594-2
- ↑ Sheng Chao, Tien; Lee, Chung Len; Lei, Tan Fu (1993), "The refractive index of InP and its oxide measured by multiple-angle incident ellipsometry", Journal of Materials Science Letters, 12 (10): 721, doi:10.1007/BF00626698.
- ↑ "Basic Parameters of InP".
- ↑ Lide, David R. (1998), Handbook of Chemistry and Physics (87 ed.), Boca Raton, Florida: CRC Press, pp. 5–20, ISBN 0-8493-0594-2
- ↑ Indium Phosphide at HSDB
- ↑ InP manufacture
- ↑ Indium Phosphide and Indium Gallium Arsenide Help Break 600 Gigahertz Speed Barrier. April 2005
- ↑ The Light Brigade appeared in Red Herring in 2002. Archived June 7, 2011, at the Wayback Machine.
- ↑ Bouarissa, Nadir (July 2011). "Phonons and related crystal properties in indium phosphide under pressure". Physica B: Condensed Matter. 406 (13): 2583–2587. doi:10.1016/j.physb.2011.03.073. Retrieved 22 March 2013.