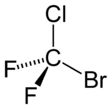
Bromochlorodifluoromethane
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
Bromo(chloro)difluoromethane | |||
| Other names
Bromochlorodifluoromethane Halon 1211 Halon 1211 BCF BCF Freon 12B1 | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.944 | ||
| EC Number | 206-537-9 | ||
PubChem CID |
|||
| UNII | |||
| |||
| |||
| Properties | |||
| CBrClF2 | |||
| Molar mass | 165.36 g/mol | ||
| Appearance | Colorless gas | ||
| Density | 1.799 g/cm3 | ||
| Melting point | −159.5 °C (−255.1 °F; 113.6 K) | ||
| Boiling point | −3.7 °C (25.3 °F; 269.4 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Bromochlorodifluoromethane, also known by the trade name Halon 1211, or BCF, or Halon 1211 BCF, or Freon 12B1, is a haloalkane with the chemical formula CF2ClBr.
Brominated haloalkanes were first used during World War II as fire extinguisher for aircraft and tanks. Bromochlorodifluoromethane was introduced as an effective gaseous fire suppression agent in the mid 1960s for use around highly valuable materials in places such as museums, mainframe rooms and telecommunication switching centers. They were also widely used in the maritime industries in the engine rooms of ships and also in the transport industry in vehicles. Its efficiency as a fire extinguishing agent has also led it to be the predominant choice of fire extinguishing agent on commercial aircraft and is typically found in cylindrical hand-held canisters. Its advantages as a fire extinguishing agent are that it has lower toxicity than chemicals such as carbon tetrachloride and that since it is a covalently bonded compound, it does not form conductive ions, therefore being usable on electrical equipment.
Use as a fire extinguishing agent, issues as an ozone depleting substance
Halon 1211 is an excellent fire extinguishing agent, as it is a streaming agent with low toxicity, a low pressure, liquefied gas, and effective on all common types of fires, A, B, and C.[1][2] It is mainly used in portable and wheeled extinguishers, and small spot protection units for marine engine applications, and was never widely used in fixed systems like Halon 1301 was.
Halon 1211 has fairly low toxicity. The lethal concentration for 15 minute exposition is about 32%.[3]
The production of bromochlorodifluoromethane and similar chlorofluorocarbons has been banned in most countries since January 1, 1994 as part of the Montreal Protocol on ozone depleting substances. However, recycling of halon 1211 allows it to remain in use, although parts availability is limited to a few manufacturers and can be an issue. Halon 1211 is still widely used in the United States, despite its high cost, with the US Military being the biggest user, but Europe and Australia have banned its use for all but "critical applications" such as aviation, military, and police use.
The manufacture of UL Listed halon 1211 extinguishers was supposed to cease on October, 2009. The future listing is still in discussion. Halotron I is the replacement extinguishing agent. It takes a larger volume to get the same ratings as 1211 has.
This is a volatile extinguishing agent that should be used only with a breathing apparatus.

Synthesis
Bromochlorodifluoromethane is commercially synthetized in a two-step way. First, chloroform is fluorinated with hydrogen fluoride. The resulting chlorodifluoromethane is then reacted with elemental bromine at 400-600 °C, with reaction time limited to about 3 seconds. The overall yield is over 90%.[3]
See also
References
- ↑ George H. Tryon et al., "Fire Protection Handbook Thirteenth Edition 1969", National Fire Protection Association, Boston Massachusetts, 1969, Library of Congress 62-12655
- ↑ Arthur E. Cote et al., "Fire Protection Handbook Eighteenth Edition", National Fire Protection Association, Quincy, Massachusetts, 1997, ISBN 0-87765-377-1
- 1 2 Rozen, Shlomo; Hagooly, Aviv (2005). "Bromochlorodifluoromethane". Encyclopedia of Reagents for Organic Synthesis. doi:10.1002/047084289X.rn00562. ISBN 0471936235.
External links
- International Chemical Safety Card 0635
- Institut national de recherche et de sécurité (1988). "Bromochlorodifluorométhane." Fiche toxicologique n° 165. Paris:INRS. (in French)
- Basic Facts about Halon
- Recycling Halon
- Aviation fire extinguisher requirements