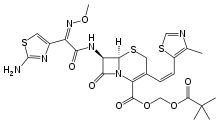
Cefditoren
 | |
| Clinical data | |
|---|---|
| Trade names | Spectracef |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a605003 |
| ATC code | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| ECHA InfoCard |
100.214.754 |
| Chemical and physical data | |
| Formula | C19H18N6O5S3 |
| Molar mass | 506.58 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
Cefditoren is a third-generation cephalosporin antibiotic for oral use. It is commonly marketed under the trade name Spectracef by Vansen Pharma Inc.
Cefditoren is also marketed under the name Meiact by Meiji Seika Pharma Co., Ltd.[1]
Spectrum of bacterial susceptibility
Cefditoren pivoxil has a broad spectrum of activity and has been used to treat bacterial infections of the skin and respiratory tract, including bronchitis, pneumonia, and tonsillitis. The following represents MIC susceptibility data for a few medically significant microorganisms.
- Haemophilus influenzae: ≥0.063 - 0.25 μg/ml
- Staphylcoccus aureus: 0.25 - >128 μg/ml (includes MRSA)
- Streptococcus pyogenes: ≤0.004 - 2 μg/ml[2]
Cefditoren is a broad-spectrum antibiotic against Gram-negative and Gram-positive bacteria, but does not have antibacterial activity against Pseudomonas aeruginosa.[3]
Clinical use
Indications
Cefditoren pivoxil is used to treat uncomplicated skin and skin structure infections, community-acquired pneumonia, acute bacterial exacerbation of chronic bronchitis, pharyngitis, and tonsillitis.
Formulations
Cefditoren pivoxil is the active ingredient in Spectracef and is available as 200- and 400-mg tablets. Cefditoren pivoxil is a prodrug which is metabolized in the body to release cefditoren as the active metabolite.
References
- ↑ Meiact Full Description Archived 2010-09-17 at the Wayback Machine.
- ↑ http://www.toku-e.com/Assets/MIC/Cefditoren%20sodium.pdf
- ↑ "Disease relevance of Cefditoren". Retrieved June 24, 2014.
https://www.accessdata.fda.gov/drugsatfda_docs/nda/2001/21-222_Spectracef.cfm