Ammonium orthomolybdate
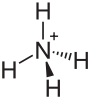 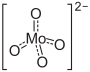 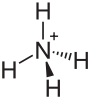 | |
| Names | |
|---|---|
| Other names
Diammonium molybdate | |
| Identifiers | |
| ChEBI | |
| ECHA InfoCard | 100.032.741 |
PubChem CID |
|
| Properties | |
| (NH4)2MoO4 | |
| Molar mass | 196.02 g/mol |
| Appearance | white solid |
| Hazards | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
1870 mg/kg (rabbit, oral) 2200 mg/kg (guinea pig, oral) 1600 mg/kg (cat, oral)[1] |
LDLo (lowest published) |
120 mg Mo/kg (rat, oral) 120 mg Mo/kg (guinea pig, oral)[1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Ammonium orthomolybdate is the inorganic compound with the chemical formula (NH4)2MoO4. It is a white solid that is prepared by treating molybdenum trioxide with aqueous ammonia. Upon heating these solutions, ammonia is lost, to give ammonium heptamolybdate ((NH4)6Mo7O24.4H2O). Ammonium orthomolybdate is used as a corrosion inhibitor and is an intermediate in some schemes to win molybdenum from its ores.[2]
Chemical reactions
Heating ammonium orthomolybdate solid or treatment with acid gives molybdenum trioxide. Such reactions proceed via ammonium dimolybdate. This equilibrium is exploited in the purification of molybdenum from its ores. Aqueous solutions of ammonium orthomolybdate react with hydrogen sulfide to give ammonium tetrathiomolybdate:
- (NH4)2MoO4 + 4 H2S → (NH4)2MoS4 + 4 H2O
References
- 1 2 "Molybdenum (soluble compounds, as Mo)". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ↑ Roger F. Sebenik et al. "Molybdenum and Molybdenum Compounds" in Ullmann's Encyclopedia of Chemical Technology 2005; Wiley-VCH, Weinheim. doi:10.1002/14356007.a16_655
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.