Acetarsol
 | |
| Names | |
|---|---|
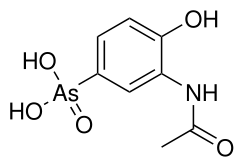
| Preferred IUPAC name
(3-Acetamido-4-hydroxyphenyl)arsonic acid | |
| Other names
3-Acetamido-4-hydroxyphenylarsonic acid Acetarsol | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.002.349 |
| EC Number | 202-582-3 |
| KEGG | |
| MeSH | Acetarsol |
PubChem CID |
|
| UNII | |
| UN number | 3465 |
| |
| |
| Properties | |
| C8H10AsNO5 | |
| Molar mass | 275.0903 g mol−1 |
| Pharmacology | |
| A07AX02 (WHO) G01AB01 (WHO), P01CD02 (WHO), P51AD05 (WHO) | |
| Hazards | |
| GHS pictograms |   |
| GHS signal word | Danger |
| H301, H331, H410 | |
| P261, P273, P301+310, P311, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Acetarsol is an anti-infective.[1]
It was first discovered in 1921 at Pasteur Institute by Ernest Fourneau, and sold under the brand name Stovarsol (fourneau is the French word for stove).[2][3]
It has been given in suppositories.[4]
Acetarsol can be used to make Arsthinol, & presumably Acetarsone also.
References
- ↑ Chen MY, Smith NA, Fox EF, Bingham JS, Barlow D (April 1999). "Acetarsol pessaries in the treatment of metronidazole resistant Trichomonas vaginalis". Int J STD AIDS. 10 (4): 277–80. doi:10.1258/0956462991913943. PMID 12035784.
- ↑ Éric Fouassier, Ces poisons qui guérissent, oct. 1996, p. 5.
- ↑ Traité de chimie organique, sous la direction de Victor Grignard, Paul Baud, vol. 22, Masson, 1959, p. 1127-1130.
- ↑ Gionchetti P, Rizzello F, Morselli C, Campieri M (October 2004). "Review article: problematic proctitis and distal colitis". Aliment. Pharmacol. Ther. 20 Suppl 4: 93–6. doi:10.1111/j.1365-2036.2004.02049.x. PMID 15352902.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.