Cobalt(II) sulfate
Cobalt(II) sulfate is any of the inorganic compounds with the formula CoSO4(H2O)x. Usually cobalt sulfate refers to the hydrate CoSO4.7H2O, which is one of the most commonly available salts of cobalt.
 | |
| Names | |
|---|---|
| IUPAC name
Cobalt(II) sulfate | |
| Other names
Colbaltous sulfate | |
| Identifiers | |
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.030.291 |
| EC Number |
|
PubChem CID |
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA) |
|
| |
| |
| Properties | |
| CoSO4·(H2O)7 | |
| Molar mass | 154.996 g/mol (anhydrous) 173.01 g/mol (monohydrate) 263.08 g/mol (hexahydrate) 281.103 g/mol (heptahydrate) |
| Appearance | reddish crystalline (anhydrous, monohydrate) pink salt (hexahydrate) |
| Odor | odorless (heptahydrate) |
| Density | 3.71 g/cm3 (anhydrous) 3.075 g/cm3 (monohydrate) 2.019 g/cm3 (hexahydrate) 1.948 g/cm3 (heptahydrate) |
| Melting point | 735 °C (1,355 °F; 1,008 K) |
| anhydrous: 36.2 g/100 mL (20 °C) 38.3 g/100 mL (25 °C) 84 g/100 mL (100 °C) heptahydrate: 60.4 g/100 mL (3 °C) 67 g/100 mL (70 °C) | |
| Solubility | anhydrous: 1.04 g/100 mL (methanol, 18 °C) insoluble in ammonia heptahydrate: 54.5 g/100 mL (methanol, 18 °C) |
| +10,000·10−6 cm3/mol | |
Refractive index (nD) |
1.639 (monohydrate) 1.540 (hexahydrate) 1.483 (heptahydrate) |
| Structure | |
| orthorhombic (anhydrous) monoclinic (monohydrate, heptahydrate) | |
| Hazards | |
| Safety data sheet | JT Baker MSDS |
EU classification (DSD) (outdated) |
Carc. Cat. 2 Muta. Cat. 3 Repr. Cat. 2 Toxic (T) Dangerous for the environment (N) |
| R-phrases (outdated) | R49, R60, R22, R42/43, R68, R50/53 |
| S-phrases (outdated) | S53, S45, S60, S61 |
| NFPA 704 (fire diamond) | |
| Flash point | Non-flammable |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
424 mg/kg (oral, rat) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
Properties, preparation, and structure
Cobalt(II) sulfate heptahydrate appears as red monoclinic crystals that liquify around 100 °C and become anhydrous at 250 °C. It is soluble in water, slightly soluble in ethanol, and especially soluble in methanol. The salts are paramagnetic.
It forms by the reaction of metallic cobalt, its oxide, hydroxide, or carbonate with aqueous sulfuric acid.[1]
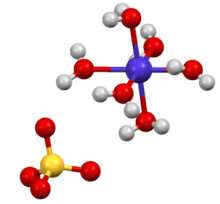
The hexahydrate is a metal aquo complex consisting of octahedral [Co(H2O)6]2+ ions associated with sulfate anions.[2]
Uses
Cobalt is obtained from ores via the sulfate in many cases.[1][3]
Hydrated cobalt(II) sulfate is used in the preparation of pigments, as well as in the manufacture of other cobalt salts. Cobalt pigment is used in porcelains and glass. Cobalt(II) sulfate is used in storage batteries and electroplating baths, sympathetic inks, and as an additive to soils and animal feeds. For these purposes, the cobalt sulfate is produced by treating cobalt oxide with sulfuric acid.[1]
Health issues
Cobalt is essential for most higher forms of life, but more than a few milligrams each day is harmful. Although poisonings have rarely resulted from cobalt compounds, their chronic ingestion has caused serious health problems at doses far less than the lethal dose. In 1966, the addition of cobalt compounds to stabilize beer foam in Canada led to a peculiar form of toxin-induced cardiomyopathy, which came to be known as beer drinker's cardiomyopathy.[4][5][6]
Furthermore, cobalt(II) sulfate is suspected of causing cancer (i.e., possibly carcinogenic, IARC Group 2B) as per the International Agency for Research on Cancer (IARC) Monographs. [PDF]
See also
References
- John D. Donaldson, Detmar Beyersmann "Cobalt and Cobalt Compounds" in Ullmann's Encyclopedia of Industrial Chemistry 2005, Wiley-VCH, Weinheim. doi:10.1002/14356007.a07_281.pub2
- Elerman, Y. "Refinement of the crystal structure of CoSO4*6H2O" Acta Crystallographica Section C 1988, volume 44, p599-p601. doi:10.1107/S0108270187012447
- Rarely, cobalt(II) sulfate is found in form of few crystallohydrate minerals, occurring among oxidation zones containing primary Co minerals (like skutterudite or cobaltite). These minerals are: biebierite (heptahydrate), moorhouseite (Co,Ni,Mn)SO4.6H2O, aplowite (Co,Mn,Ni)SO4.4H2O and cobaltkieserite (monohydrate).
- Morin Y; Tětu A; Mercier G (1969). "Quebec beer-drinkers' cardiomyopathy: Clinical and hemodynamic aspects". Annals of the New York Academy of Sciences. 156 (1): 566–576. Bibcode:1969NYASA.156..566M. doi:10.1111/j.1749-6632.1969.tb16751.x. PMID 5291148.
- Barceloux, Donald G. & Barceloux, Donald (1999). "Cobalt". Clinical Toxicology. 37 (2): 201–216. doi:10.1081/CLT-100102420. PMID 10382556.
- 11.1.5 The unusual type of myocardiopathy recognized in 1965 and 1966 in Quebec (Canada), Minneapolis (Minnesota), Leuven (Belgium), and Omaha (Nebraska) was associated with episodes of acute heart failure (e/g/, 50 deaths among 112 beer drinkers).
