Triethylamine
| |||
| Names | |||
|---|---|---|---|
| Preferred IUPAC name
N,N-Diethylethanamine | |||
| Other names
(Triethyl)amine Triethylamine (deprecated[1]) | |||
| Identifiers | |||
3D model (JSmol) |
|||
| Abbreviations | TEA[2] | ||
| 605283 | |||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.004.064 | ||
| EC Number | 204-469-4 | ||
| KEGG | |||
| MeSH | triethylamine | ||
PubChem CID |
|||
| RTECS number | YE0175000 | ||
| UNII | |||
| UN number | 1296 | ||
| |||
| |||
| Properties[3] | |||
| C6H15N | |||
| Molar mass | 101.19 g·mol−1 | ||
| Appearance | Colourless liquid | ||
| Odor | Fishy, ammoniacal | ||
| Density | 0.7255 g mL−1 | ||
| Melting point | −114.70 °C; −174.46 °F; 158.45 K | ||
| Boiling point | 88.6 to 89.8 °C; 191.4 to 193.5 °F; 361.7 to 362.9 K | ||
| log P | 1.647 | ||
| Vapor pressure | 6.899–8.506 kPa | ||
Henry's law constant (kH) |
66 μmol Pa−1 kg−1 | ||
| Acidity (pKa) | 10.75 (for the conjugate acid) (H2O), 9.00 (DMSO) [4] | ||
| -81.4·10−6 cm3/mol | |||
Refractive index (nD) |
1.401 | ||
| Thermochemistry | |||
Heat capacity (C) |
216.43 J K−1 mol−1 | ||
Std enthalpy of formation (ΔfH |
−169 kJ mol−1 | ||
Std enthalpy of combustion (ΔcH |
−4.37763 to −4.37655 MJ mol−1 | ||
| Hazards | |||
| GHS pictograms |  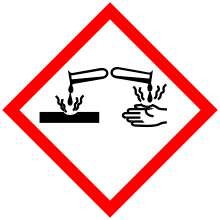  | ||
| GHS signal word | DANGER | ||
| H225, H302, H312, H314, H332 | |||
| P210, P280, P305+351+338, P310 | |||
| NFPA 704 | |||
| Flash point | −15 °C (5 °F; 258 K) | ||
| 312 °C (594 °F; 585 K) | |||
| Explosive limits | 1.2–8% | ||
| 2 ppm (8 mg/m3) (TWA), 4 ppm (17 mg/m3) (STEL) | |||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose) |
| ||
LCLo (lowest published) |
1425 ppm (mouse, 2 hr)[5] | ||
| US health exposure limits (NIOSH): | |||
PEL (Permissible) |
TWA 25 ppm (100 mg/m3)[6] | ||
REL (Recommended) |
None established[6] | ||
IDLH (Immediate danger) |
200 ppm[6] | ||
| Related compounds | |||
Related amines |
|||
Related compounds |
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Triethylamine is the chemical compound with the formula N(CH2CH3)3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine or tetraethylammonium, for which TEA is also a common abbreviation. It is a colourless volatile liquid with a strong fishy odor reminiscent of ammonia and is also the smell of the hawthorn plant.[7] Like diisopropylethylamine (Hünig’s base), triethylamine is commonly employed in organic synthesis.
Synthesis and properties
Triethylamine is prepared by the alkylation of ammonia with ethanol:[8]
- NH3 + 3 C2H5OH → N(C2H5)3 + 3 H2O
The pKa of protonated triethylamine is 10.75,[4] and it can be used to prepare buffer solutions at that pH. The hydrochloride salt, triethylamine hydrochloride (triethylammonium chloride), is a colorless, odorless, and hygroscopic powder, which decomposes when heated to 261 °C.
Laboratory samples of triethylamine can be purified by distilling from calcium hydride.[9]
Applications
Triethylamine is commonly employed in organic synthesis as a base. For example, it is commonly used as a base during the preparation of esters and amides from acyl chlorides.[10] Such reactions lead to the production of hydrogen chloride which combines with triethylamine to form the salt triethylamine hydrochloride, commonly called triethylammonium chloride. This reaction removes the hydrogen chloride from the reaction mixture, which can be required for these reactions to proceed to completion (R, R' = alkyl, aryl):
- R2NH + R'C(O)Cl + Et3N → R'C(O)NR2 + Et3NH+Cl−
Like other tertiary amines, it catalyzes the formation of urethane foams and epoxy resins. It is also useful in dehydrohalogenation reactions and Swern oxidations.
Triethylamine is readily alkylated to give the corresponding quaternary ammonium salt:
- RI + Et3N → Et3NR+I−
Triethylamine is mainly used in the production of quaternary ammonium compounds for textile auxiliaries and quaternary ammonium salts of dyes. It is also a catalyst and acid neutralizer for condensation reactions and is useful as an intermediate for manufacturing medicines, pesticides and other chemicals.
Niche uses
Triethylamine is used to give salts of various carboxylic acid-containing pesticides, e.g. Triclopyr and 2,4-dichlorophenoxyacetic acid
Triethylamine is the active ingredient in FlyNap, a product for anesthetizing Drosophila melanogaster. Triethylamine is used in mosquito and vector control labs to anesthetize mosquitoes. This is done to preserve any viral material that might be present during species identification.
Also, the bicarbonate salt of triethylamine (often abbreviated TEAB, triethylammonium bicarbonate) is useful in reverse phase chromatography, often in a gradient to purify nucleotides and other biomolecules.
References
- ↑ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 671. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ↑ X. Bories-Azeau, S. P. Armes, and H. J. W. van den Haak, Macromolecules 2004, 37, 2348 PDF
- ↑ The Merck Index (11th ed.). 9582.
- 1 2 David Evans Research Group Archived 2012-01-21 at the Wayback Machine.
- ↑ "Triethylamine". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- 1 2 3 "NIOSH Pocket Guide to Chemical Hazards #0633". National Institute for Occupational Safety and Health (NIOSH).
- ↑ The Hawthorn, BBC
- ↑ Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke "Amines, Aliphatic" in Ullmann's Encyclopedia of Industrial Chemistry, Wiley-VCH, Weinheim, 2005. doi:10.1002/14356007.a02_001
- ↑ F.,, Armarego, W. L. Purification of Laboratory Chemicals. Chai, Christina Li Lin, (Seventh ed.). Amsterdam. ISBN 9780123821621. OCLC 820853648.
- ↑ Sorgi, K. L. (2001). "Triethylamine". Encyclopedia of Reagents for Organic Synthesis. Encyclopedia of Reagents for Organic Synthesis. New York: John Wiley & Sons. doi:10.1002/047084289X.rt217. ISBN 0471936235.