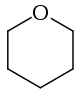
Tetrahydropyran
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Oxane | |||
| Other names
Tetrahydropyran, Oxacyclohexane | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChEBI | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.005.048 | ||
| KEGG | |||
PubChem CID |
|||
| UNII | |||
| |||
| |||
| Properties | |||
| C5H10O | |||
| Molar mass | 86.13 g·mol−1 | ||
| Density | 0.880 g/cm3 | ||
| Melting point | −45 °C (−49 °F; 228 K) | ||
| Boiling point | 88 °C (190 °F; 361 K) | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Tetrahydropyran (THP) is the organic compound consisting of a saturated six-membered ring containing five carbon atoms and one oxygen atom. It is named by reference to pyran, which contains two double bonds, and may be produced from it by adding four hydrogens. In 2013, its preferred IUPAC name was established as oxane.[1] The compound is a colourless volatile liquid, but is obscure. Derivatives of tetrahydropyran are, however, more common. 2-Tetrahydropyranyl (THP-) ethers derived from the reaction of alcohols and 3,4-dihydropyran are commonly used as protecting groups in organic synthesis.[2] Furthermore, a tetrahydropyran ring system, i.e., five carbon atoms and an oxygen, is the core of pyranose sugars, such as glucose. In gas phase, the THP exists in its lowest energy Cs symmetry chair conformation.[3]
Preparation
One classic procedure for the organic synthesis of tetrahydropyran is by hydrogenation of the 3,4-isomer of dihydropyran with Raney nickel.[4]
Reactions
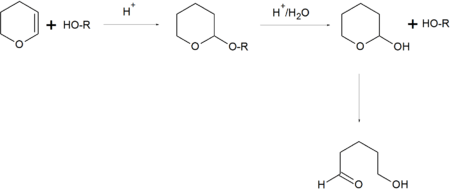
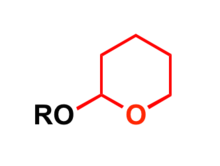
In organic synthesis, the 2-tetrahydropyranyl group is used as a protecting group for alcohols.[5][6] Reaction of the alcohol with 3,4-dihydropyran forms a 2-tetrahydropyranyl ether, protecting the alcohol from a variety of reactions. The alcohol can later be restored readily by acidic hydrolysis with formation of 5-hydroxypentanal.[2]
Alcohol protection
In organic synthesis, 2-tetrahydropyranyl group (THP) is used as a protecting group for alcohols by the formation of THP ethers.
Most common protection methods
- Treatment of alcohol with 3,4-dihydropyran and p-toluenesulfonic acid in dichloromethane at ambient temperature[2]

- 2-hydroxytetrahydropyranyl, triphenylphosphine, diethyl azodicarboxylate (DEAD) in tetrahydrofuran (THF)
Most common deprotection methods
- Acetic acid (AcOH) in THF/water solution or p-toluenesulfonic acid in water
- Pyridinium p-toluenesulfonate (PPTS) in ethanol
See also
References
- ↑ "New IUPAC Organic Nomenclature - Chemical Information BULLETIN" (PDF).
- 1 2 3 Wuts, Peter G. M.; Greene, Theodora W. (2006). "Protection for the Hydroxyl Group, Including 1,2‐ and 1,3‐Diols". Greene's Protective Groups in Organic Synthesis (4th ed.). pp. 16–366. doi:10.1002/9780470053485.ch2. ISBN 9780470053485.
- ↑ Builth-Williams, J. D.; Bellm, S. M.; Chiari, L.; Thorn, P. A.; Jones, D. B.; Chaluvadi, H.; Madison, D. H.; Ning, C. G.; Lohmann, B. (2013). "A dynamical (e,2e) investigation of the structurally related cyclic ethers tetrahydrofuran, tetrahydropyran, and 1,4-dioxane". Journal of Chemical Physics. 139 (3): 034306. doi:10.1063/1.4813237.
- ↑ Andrus, D. W.; Johnson, John R. (1943). "Tetrahydropyran". Organic Syntheses. 23: 90. doi:10.15227/orgsyn.023.0090. ; Collective Volume, 3, p. 794
- ↑ Earl, R. A.; Townsend, L. B. (1981). "Methyl 4-Hydroxy-2-butynoate". Organic Syntheses. 60: 81. doi:10.15227/orgsyn.060.0081. ; Collective Volume, 7, p. 334
- ↑ Kluge, Arthur F. (1986). "Diethyl [(2-Tetrahydropyranyloxy)methyl]phosphonate". Organic Syntheses. 64: 80. doi:10.15227/orgsyn.064.0080. ; Collective Volume, 7, p. 160
- ↑ Robinson, Anna; Aggarwal, Varinder K. (2010). "Asymmetric Total Synthesis of Solandelactone E: Stereocontrolled Synthesis of the 2-ene-1,4-diol Core through a Lithiation–Borylation–Allylation Sequence". Angewandte Chemie International Edition. 49 (37): 6673–6675. doi:10.1002/anie.201003236.