Talnetant
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| ChemSpider | |
| UNII | |
| ECHA InfoCard |
100.236.526 |
| Chemical and physical data | |
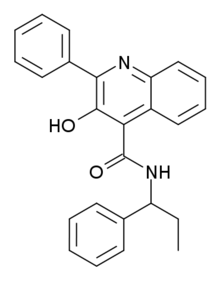
| Formula | C25H22N2O2 |
| Molar mass | 382.454 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
Talnetant (SB-223,412) is a neurokinin 3 receptor antagonist developed by GlaxoSmithKline, which is being researched for several functions (primarily treatment of irritable bowel syndrome, despite a 2007 study finding no statistically significant improvement in rectal hypersensitivity over placebo). Its use as a potential antipsychotic drug for the treatment of schizophrenia has also been discontinued.[1][2][3][4]
See also
References
- ↑ "UPDATE 1-GlaxoSmithKline prunes new drug pipeline". Reuters. Oct 15, 2007.
- ↑ Evangelista S. Talnetant GlaxoSmithKline. Current Opinion on Investigational Drugs. 2005 Jul;6(7):717-21. PMID 16044668
- ↑ Houghton LA, Cremonini F, Camilleri M, Busciglio I, Fell C, Cox V, Alpers DH, Dewit OE, Dukes GE, Gray E, Lea R, Zinsmeister AR, Whorwell PJ. Effect of the NK(3) receptor antagonist, talnetant, on rectal sensory function and compliance in healthy humans. Neurogastroenterology and Motility. 2007 Sep;19(9):732-43. PMID 17727393
- ↑ Dawson LA, Cato KJ, Scott C, Watson JM, Wood MD, Foxton R, de la Flor R, Jones GA, Kew JN, Cluderay JE, Southam E, Murkitt GS, Gartlon J, Pemberton DJ, Jones DN, Davies CH, Hagan J. In Vitro and In Vivo Characterization of the Non-peptide NK(3) Receptor Antagonist SB-223412 (Talnetant): Potential Therapeutic Utility in the Treatment of Schizophrenia. Neuropsychopharmacology. 2008 Jun;33(7):1642-52. PMID 17728699
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.