Phosphorus trichloride
 | |
 | |
 | |
| Names | |
|---|---|
| IUPAC name
Phosphorus trichloride | |
| Other names
Phosphorus(III) chloride Phosphorous chloride | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.028.864 |
| EC Number | 231-749-3 |
PubChem CID |
|
| RTECS number | TH3675000 |
| UNII | |
| UN number | 1809 |
| |
| |
| Properties | |
| PCl3 | |
| Molar mass | 137.33 g/mol |
| Appearance | Colorless to yellow fuming liquid[1] |
| Odor | like hydrochloric acid[1] |
| Density | 1.574 g/cm3 |
| Melting point | −93.6 °C (−136.5 °F; 179.6 K) |
| Boiling point | 76.1 °C (169.0 °F; 349.2 K) |
| hydrolysis | |
| Solubility in other solvents | soluble in benzene, CS2, ether, chloroform, CCl4, halogenated organic solvents reacts with ethanol |
| Vapor pressure | 13.3 kPa |
| −63.4·10−6 cm3/mol | |
Refractive index (nD) |
1.5122 (21 °C) |
| Viscosity | 0.65 cP (0 °C) 0.438 cP (50 °C) |
| 0.97 D | |
| Thermochemistry | |
Std enthalpy of formation (ΔfH |
−319.7 kJ/mol |
| Hazards | |
| Safety data sheet | See: data page ICSC 0696 |
EU classification (DSD) (outdated) |
|
| R-phrases (outdated) | R14-R26/28-R35-R48/20 |
| S-phrases (outdated) | (S1/2)-S7/8-S26-S36/37/39-S45 |
| NFPA 704 | |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
18 mg/kg (rat, oral)[2] |
LC50 (median concentration) |
104 ppm (rat, 4 hr) 50 ppm (guinea pig, 4 hr)[2] |
| US health exposure limits (NIOSH): | |
PEL (Permissible) |
TWA 0.5 ppm (3 mg/m3)[1] |
REL (Recommended) |
TWA 0.2 ppm (1.5 mg/m3) ST 0.5 ppm (3 mg/m3)[1] |
IDLH (Immediate danger) |
25 ppm[1] |
| Related compounds | |
Related phosphorus chlorides |
Phosphorus pentachloride Phosphorus oxychloride Diphosphorus tetrachloride |
Related compounds |
Phosphorus trifluoride Phosphorus tribromide Phosphorus triiodide |
| Supplementary data page | |
| Refractive index (n), Dielectric constant (εr), etc. | |
Thermodynamic data |
Phase behaviour solid–liquid–gas |
| UV, IR, NMR, MS | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
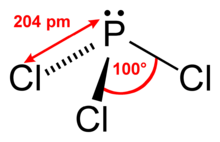
Phosphorus trichloride is a chemical compound of phosphorus and chlorine, having the chemical formula PCl3. It has a trigonal pyramidal shape. It is the most important of the three phosphorus chlorides. It is an important industrial chemical, being used for the manufacture of organophosphorus compounds for a wide variety of applications. It has a 31P NMR signal at around +220 ppm with reference to a phosphoric acid standard.
Chemical properties
The phosphorus in PCl3 is often considered to have the +3 oxidation state and the chlorine atoms are considered to be in the −1 oxidation state. Most of its reactivity is consistent with this description.
Redox reactions
PCl3 is a precursor to other phosphorus compounds, undergoing oxidation to phosphorus pentachloride (PCl5), thiophosphoryl chloride (PSCl3), or phosphorus oxychloride (POCl3).
If an electric discharge is passed through a mixture of PCl3 vapour and hydrogen gas, a rare chloride of phosphorus is formed, diphosphorus tetrachloride (P2Cl4).
PCl3 as an electrophile
Phosphorus trichloride is the precursor to organophosphorus compounds that contain one or more P(III) atoms, most notably phosphites and phosphonates. These compounds do not usually contain the chlorine atoms found in PCl3.
PCl3 reacts vigorously with water to form phosphorous acid, H3PO3 and HCl:
- PCl3 + 3 H2O → H3PO3 + 3 HCl
A large number of similar substitution reactions are known, the most important of which is the formation of phosphites by reaction with alcohols or phenols. For example, with phenol, triphenyl phosphite is formed:
- 3 PhOH + PCl3 → P(OPh)3 + 3 HCl
where "Ph" stands for phenyl group, -C6H5. Alcohols such as ethanol react similarly in the presence of a base such as a tertiary amine:[3]
- PCl3 + 3 EtOH + 3 R3N → P(OEt)3 + 3 R3NH+Cl−
In the absence of base, however, the reaction proceeds with the following stoichiometry to give diethylphosphite:[4][5]
- PCl3 + 3 EtOH → (EtO)2P(O)H + 2 HCl + EtCl
Secondary amines (R2NH) form aminophosphines, e.g., tris(dimethylamino)phosphine. Thiols (RSH) form P(SR)3. An industrially relevant reaction of PCl3 with amines is phosphonomethylation, which employs formaldehyde:
- R2NH + PCl3 + CH2O → (HO)2P(O)CH2NR2 + 3 HCl
Aminophosphonates are widely used as sequestring and antiscale agents in water treatment. The large volume herbicide glyphosate is also produced this way. The reaction of PCl3 with Grignard reagents and organolithium reagents is a useful method for the preparation of organic phosphines with the formula R3P (sometimes called phosphanes) such as triphenylphosphine, Ph3P.
- 3 PhMgBr + PCl3 → Ph3P + 3 MgBrCl
Under controlled conditions or especially with bulky organic groups, similar reactions afford less substituted derivatives such as chlorodiisopropylphosphine.
PCl3 as a nucleophile
Phosphorus trichloride has a lone pair, and therefore can act as a Lewis base,[6] e.g., forming a 1:1 adduct Br3B-PCl3. Metal complexes such as Ni(PCl3)4 are known, again demonstrating the ligand properties of PCl3.
This Lewis basicity is exploited in the Kinnear–Perren reaction to prepare alkylphosphonyl dichlorides (RP(O)Cl2) and alkylphosphonate esters (RP(O)(OR')2). Alkylation of phosphorus trichloride is effected in the presence of aluminium trichloride give the alkyltrichlorophosphonium salts, which are versatile intermediates:[7]
- PCl3 + RCl + AlCl3 → RPCl+
3 + AlCl−
4
The RPCl+
3 product can then be decomposed with water to produce an alkylphosphonic dichloride RP(=O)Cl2.
Preparation
World production exceeds one-third of a million tonnes.[8] Phosphorus trichloride is prepared industrially by the reaction of chlorine with a refluxing solution of white phosphorus in phosphorus trichloride, with continuous removal of PCl3 as it is formed (in order to avoid the formation of PCl5).
- P4 + 6 Cl2 → 4 PCl3
Industrial production of phosphorus trichloride is controlled under the Chemical Weapons Convention, where it is listed in schedule 3. In the laboratory it may be more convenient to use the less toxic red phosphorus.[9] It is sufficiently inexpensive that it would not be synthesized for laboratory use.
Uses
PCl3 is important indirectly as a precursor to PCl5, POCl3 and PSCl3, which are used in many applications, including herbicides, insecticides, plasticisers, oil additives, and flame retardants.
For example, oxidation of PCl3 gives POCl3, which is used for the manufacture of triphenyl phosphate and tricresyl phosphate, which find application as flame retardants and plasticisers for PVC. They are also used to make insecticides such as diazinon. Phosphonates include the herbicide glyphosate.
PCl3 is the precursor to triphenylphosphine for the Wittig reaction, and phosphite esters which may be used as industrial intermediates, or used in the Horner-Wadsworth-Emmons reaction, both important methods for making alkenes. It can be used to make trioctylphosphine oxide (TOPO), used as an extraction agent, although TOPO is usually made via the corresponding phosphine.
PCl3 is also used directly as a reagent in organic synthesis. It is used to convert primary and secondary alcohols into alkyl chlorides, or carboxylic acids into acyl chlorides, although thionyl chloride generally gives better yields than PCl3.[10]
Precautions
PCl3 is toxic, with a concentration of 600 ppm being lethal in just a few minutes.[11] PCl3 is classified as very toxic and corrosive under EU Directive 67/548/EEC, and the risk phrases R14, R26/28, R35 and R48/20 are obligatory.
Government agencies in the United States have set occupational exposure limits for PCl3. The Occupational Safety and Health Administration has set a permissible exposure limit at 0.5 ppm over a time-weighted average of 8 hours, while the National Institute for Occupational Safety and Health has set a recommended exposure limit at 0.2 ppm over a time-weighted average of 8 hours.[12] Additionally, PCl3 has been designated IDLH with a maximum exposure limit at 25 ppm.[13]
History
Phosphorus trichloride was first prepared in 1808 by the French chemists Joseph Louis Gay-Lussac and Louis Jacques Thénard by heating calomel (Hg2Cl2) with phosphorus.[14] Later during the same year, the English chemist Humphry Davy produced phosphorus trichloride by burning phosphorus in chlorine gas.[15]
References
- 1 2 3 4 5 "NIOSH Pocket Guide to Chemical Hazards #0511". National Institute for Occupational Safety and Health (NIOSH).
- 1 2 "Phosphorus trichloride". Immediately Dangerous to Life and Health Concentrations (IDLH). National Institute for Occupational Safety and Health (NIOSH).
- ↑ A. H. Ford-Moore and B. J. Perry (1963). "Triethyl Phosphite". Organic Syntheses. ; Collective Volume, 4, p. 955
- ↑ Malowan, John E. (1953). "Diethyl phosphite". Inorganic Syntheses. 4: 58–60. doi:10.1002/9780470132357.ch19.
- ↑ Pedrosa, Leandro (2011). "Esterification of Phosphorus Trichloride with Alcohols; Diisopropyl phosphonate". ChemSpider Synthetic Pages. Royal Society of Chemistry: 488. doi:10.1039/SP488.
- ↑ R. R. Holmes (1960). "An examination of the basic nature of the trihalides of phosphorus, arsenic and antimony,". Journal of Inorganic and Nuclear Chemistry. 12 (3–4): 266–275. doi:10.1016/0022-1902(60)80372-7.
- ↑ Svara, J.; Weferling, N.; Hofmann, T., "Phosphorus Compounds, Organic", Ullmann's Encyclopedia of Industrial Chemistry, Weinheim: Wiley-VCH, doi:10.1002/14356007.a19_545.pub2
- ↑ Greenwood, Norman N.; Earnshaw, Alan (1997). Chemistry of the Elements (2nd ed.). Butterworth-Heinemann. ISBN 0-08-037941-9.
- ↑ M. C. Forbes; C. A. Roswell; R. N. Maxson (1946). "Phosphorus(III) Chloride". Inorg. Synth. 2: 145–7. doi:10.1002/9780470132333.ch42.
- ↑ L. G. Wade, Jr. (2005). Organic Chemistry (6th ed.). Upper Saddle River, New Jersey, USA: Pearson/Prentice Hall. p. 477.
- ↑ A. D. F. Toy (1973). The Chemistry of Phosphorus. Oxford, UK: Pergamon Press.
- ↑ CDC - NIOSH Pocket Guide to Chemical Hazards
- ↑ Documentation for Immediately Dangerous To Life or Health Concentrations (IDLHs)
- ↑ Gay-Lussac; Thénard (27 May 1808). "Extrait de plusieurs notes sur les métaux de la potasse et de la soude, lues à l'Institut depuis le 12 janvier jusqu'au 16 mai" [Extracts from several notes on the metals potassium and sodium, read at the Institute from the 12th of January to the 16th of May]. Gazette Nationale, ou le Moniteur Universel (in French). 40 (148): 581–582. From p. 582: "Seulement ils ont rapporté qu'en traitant le mercure doux par le phosphure, dans l'espérance d'avoir de l'acide muriatique bien sec, il ont trouvé une liqueur nouvelle très limpide, sans couleur, répandant de fortes vapeurs, s'enflammant spontanément lorsqu'on en imbibe le papier joseph; laquelle ne paraît être qu'une combinaison de phosphore, d'oxigène et d'acide muriatique, et par conséquent analogue à cette qu'on obtient en traitant le soufre par le gas acide muriatique oxigèné." (Only they reported that by treating calomel with phosphorus, in the hope of obtaining very dry hydrogen chloride, they found a new, very clear liquid, colorless, giving off strong vapors, spontaneously igniting when one soaks filter paper in it; which seems to be only a compound of phosphorus, oxygen, and hydrochloric acid, and thus analogous to what one obtains by treating sulfur with chlorine gas.)
- ↑ Davy, Humphry (1809). "The Bakerian Lecture. An account of some new analytical researches on the nature of certain bodies, particularly the alkalies, phosphorus, sulphur, carbonaceous matter, and the acids hitherto undecomposed; with some general observations on chemical theory". Philosophical Transactions of the Royal Society of London. 99: 39–104. On pp. 94–95, Davy mentioned that when he burned phosphorus in chlorine gas ("oxymuriatic acid gas"), he obtained a clear liquid (phosphorus trichloride) and a white solid (phosphorus pentachloride).
