Oxybenzone
 | |
| Names | |
|---|---|
| IUPAC name
(2-Hydroxy-4-methoxyphenyl)-phenylmethanone | |
| Other names
Oxybenzone Benzophenone-3 2-Hydroxy-4-methoxybenzophenone | |
| Identifiers | |
3D model (JSmol) |
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.004.575 |
| KEGG | |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C14H12O3 | |
| Molar mass | 228.25 g·mol−1 |
| Density | 1.20 g cm−3[2] |
| Melting point | 62 to 65 °C (144 to 149 °F; 335 to 338 K) |
| Boiling point | 224 to 227 °C (435 to 441 °F; 497 to 500 K) |
| Acidity (pKa) | 7.6 (H2O)[3] |
| Hazards[2] | |
| NFPA 704 | |
| Flash point | 140.5 °C (284.9 °F; 413.6 K) |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose) |
>12800 mg/kg (oral in rats) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
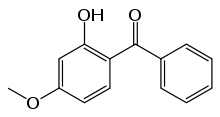
Oxybenzone or benzophenone-3 (trade names Milestab 9, Eusolex 4360, Escalol 567, KAHSCREEN BZ-3) is an organic compound. It is a pale-yellow solid that is readily soluble in most organic solvents. Oxybenzone belongs to the class of aromatic ketones known as benzophenones. It is a component of many sunscreen lotions and is added to plastics to limit UV degradation.
Structure and electronic structure
Being a conjugated molecule, oxybenzone absorbs at lower energies than many aromatic molecules.[4] As in related compounds, the hydroxyl group is hydrogen bonded to the ketone.[5] This interaction contributes to oxybenzone's light-absorption properties. At low temperatures, however, it is possible to observe both the phosphorescence and the triplet-triplet absorption spectrum. At 175 K the triplet lifetime is 24 ns. The short lifetime has been attributed to a fast intramolecular hydrogen transfer between the oxygen of the C=O and the OH.[6]
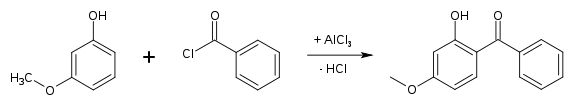
Production
Oxybenzone is produced by Friedel-Crafts reaction of benzoyl chloride with 3-methoxyphenol.[7]
Uses
Oxybenzone is used in plastics as an ultraviolet light absorber and stabilizer.[7] It is used, along with other benzophenones, in sunscreens, hair sprays, and cosmetics because they help prevent potential damage from sunlight exposure. It is also found, in concentrations up to 1%, in nail polishes.[7] Oxybenzone can also be used as a photostabilizer for synthetic resins.[7] Benzophenones can leach from food packaging, and are widely used as photo-initiators to activate a chemical that dries ink faster.[8]
As a sunscreen, it provides broad-spectrum ultraviolet coverage, including UVB and short-wave UVA rays. As a photoprotective agent, it has an absorption profile spanning from 270 to 350 nm with absorption peaks at 288 and 350 nm.[9] It is one of the most widely used organic UVA filters in sunscreens today.[9] It is also found in nail polish, fragrances, hairspray, and cosmetics as a photostabilizer. Despite its photoprotective qualities, much controversy surrounds oxybenzone because of its possible hormonal and photoallergenic effects, leading many countries to regulate its use.
Safety
Some debate focuses on the potential of oxybenzone as a contact allergen.[9] Due to the advent of PABA-free sunscreens, oxybenzone is now the most common allergen found in sunscreens.[10][11][12][13]
In vivo studies
Among common sunscreen chemicals, oxybenzone is associated with allergic reactions triggered by sun exposure. In a study of 82 patients with photoallergic contact dermatitis, over one quarter showed photoallergic reactions to oxybenzone.[14]
In a 2008 study of participants ages 6 and up, oxybenzone was detected in 96.8% of urine samples.[15] Humans can absorb anywhere from 0.4% to 8.7% of oxybenzone after one topical application of sunscreen, as measured in urine excretions. This number can increase after multiple applications over the same period of time.[16] Oxybenzone is particularly penetrative because it is the most lipophilic of the three most common UV filters.[17]
When applied topically, UV filters, such as oxybenzone, are absorbed through the skin, metabolized, and excreted primarily through the urine.[18] The method of biotransformation, the process by which a foreign compound is chemically transformed to form a metabolite, was determined by Okereke and colleagues through oral and dermal administration of oxybenzone to rats. The scientists analyzed blood, urine, feces, and tissue samples and found three metabolites: 2,4-dihydroxybenzophenone(DHB), 2,2-dihydroxy-4-methoxybenzophenone (DHMB) and 2,3,4-trihydroxybenzophenone (THB).[19][20] To form DHB the methoxy functional group undergoes o-dealkylation; to form THB the same ring is hydroxylated.[18] Ring B in oxybenzone is hydroxylated to form DHMB.[18]
A study done in 2004 measured the levels of oxybenzone and its metabolites in urine. After topical application to human volunteers, results revealed that up to 1% of the applied dose was found in the urine.[21] The major metabolite detected was DHB and very small amounts of THB were found.[21] By utilizing the Ames test in Salmonella typhimurium strains, DHB was determined to be nonmutagenic.[22]
Toxicity to coral
Benzophenones in sunscreens have been linked to coral bleaching.[23][24] A 2015 study published in the Archives of Environmental Contamination and Toxicology directly linked oxybenzone to the declining health of coral reefs.[25]
Regulation
Australia
Revised as of 2007, the National Industrial Chemicals Notification and Assessment Scheme (NICNAS) Cosmetic Guidelines allow oxybenzone for cosmetic use up to 10%.[26]
Canada
Revised as of 2012, Health Canada allows oxybenzone for cosmetic use up to 6%.[27]
European Union
The Scientific Committee on Consumer Products (SCCP) of the European Commission concluded in 2008 that it does not pose a significant risk to consumers, apart from contact allergenic potential.[28] It is allowed in sunscreens and cosmetics at levels of up to 6% and 0.5% respectively.[29]
Japan
Revised as of 2001, the Ministry of Health, Labour, and Welfare notification allows oxybenzone for cosmetic use up to 5%.[30]
Sweden
The Swedish Research Council has determined that sunscreens with oxybenzone are unsuitable for use in young children, because children under the age of two years have not fully developed the enzymes that are believed break it down. No regulations have come of this study yet.[7]
United States
Oxybenzone was approved for use in the US by the FDA in the early 1980s. Revised as of April 1, 2013, the FDA allows oxybenzone in OTC sunscreen products up to 6%.[31]
The Hawaii State Legislature has passed a bill that would prohibit the sale of non-prescription sunscreens containing oxybenzone and other chemicals damaging to coral reefs (e.g. octyl methoxycinnamate), effective January 1, 2021.[32]
References
- ↑ Merck Index, 11th Edition, 6907
- 1 2 131-57-7 at commonchemistry.org
- ↑ Fontanals, Núria; Cormack, Peter A.G.; Sherrington, David C.; Marcé, Rosa M.; Borrull, Francesc (2010). "Weak anion-exchange hypercrosslinked sorbent in on-line solid-phase extraction–liquid chromatography coupling to achieve automated determination with an effective clean-up". Journal of Chromatography A. 1217 (17): 2855–61. doi:10.1016/j.chroma.2010.02.064. PMID 20303088.
- ↑ Castro, G. T.; Blanco, S. E.; Giordano, O. S. (2000). "UV Spectral Properties of Benzophenone. Influence of Solvents and Substituents". Molecules. 5 (3): 424. doi:10.3390/50300424.
- ↑ Lago, A. F.; Jimenez, P.; Herrero, R.; Dávalos, J. Z.; Abboud, J.-L. M. (2008). "Thermochemistry and Gas-Phase Ion Energetics of 2-Hydroxy-4-methoxy-benzophenone (Oxybenzone)". The Journal of Physical Chemistry A. 112 (14): 3201–8. Bibcode:2008JPCA..112.3201L. doi:10.1021/jp7111999. PMID 18341312.
- ↑ ChréTien, Michelle N.; Heafey, Eve; Scaiano, Juan C. (2010). "Reducing Adverse Effects from UV Sunscreens by Zeolite Encapsulation: Comparison of Oxybenzone in Solution and in Zeolites". Photochemistry and Photobiology. 86 (1): 153–61. doi:10.1111/j.1751-1097.2009.00644.x. PMID 19930122.
- 1 2 3 4 5 "Hazardous Substances Data Bank". 2-HYDROXY-4-METHOXYBENZOPHENONE. National Library of Medicine (US), Division of Specialized Information Services. Retrieved 9 March 2014.
- ↑ Koivikko, R; Pastorelli, S; Rodríguez-Bernaldo de Quirós, A; Paseiro-Cerrato, R; Paseiro-Losada, P; Simoneau, C (2010). “Rapid multi-analye quantification of benzophenone, 4-methylbenzophenone and related derivatives form paperboard food packaging”. Food Addit Contam Part A Chem Anal Control Expo Risk Assess. 2010 Oct;27(10):1478-86. doi: 10.1080/19440049.2010.502130. Retrieved 8 February 2015. PMID 20640959.
- 1 2 3 Burnett, M. E.; Wang, S. Q. (2011). "Current sunscreen controversies: A critical review". Photodermatology, Photoimmunology & Photomedicine. 27 (2): 58–67. doi:10.1111/j.1600-0781.2011.00557.x. PMID 21392107.
- ↑ Rietschel, Robert L.; Fowler, Joseph F. (2008). Fisher's Contact Dermatitis (6th ed.). Hamilton: PMPH-USA. p. 460. ISBN 9781550093780. Retrieved 8 February 2015.
- ↑ DeLeo, Vincent A.; Suarez, Sylvia M.; Maso, Martha J. (1992). "Photoallergic contact dermatitis; results of photo patch testing in New York, 1985-1990". Arch Dermatol. 128 (11): 1513–1518. doi:10.1001/archderm.1992.01680210091015. Retrieved 8 February 2015.
- ↑ Scheuer, Elyse; Warshaw, Erin (March 2006). "Sunscreen Allergy: A Review of Epidemiology, Clinical Characteristics, and Responsible Allergens". Dermatitis. 17 (1): 3–11. doi:10.2310/6620.2006.05017. Retrieved 8 February 2015.
- ↑ Zhang, Xiao-Min; Nakagawa, Mikio; Kawai, Keiichi; Kawai, Kyozo (January 1998). "Erythema-multiforme-like eruption following photoallergic contact dermatitis from oxybenzone". Contact Dermatitis. 38 (1): 43–44. doi:10.1111/j.1600-0536.1998.tb05637.x. Retrieved 8 February 2015.
- ↑ Rodríguez, E; Valbuena, M. C.; Rey, M; Porras De Quintana, L (2006). "Causal agents of photoallergic contact dermatitis diagnosed in the national institute of dermatology of Colombia". Photodermatology, Photoimmunology & Photomedicine. 22 (4): 189–92. doi:10.1111/j.1600-0781.2006.00212.x. PMID 16869867.
- ↑ Calafat, A. M.; Wong, L. Y.; Ye, X.; Reidy, J. A.; Needham, L. L. (2008). "Concentrations of the Sunscreen Agent Benzophenone-3 in Residents of the United States: National Health and Nutrition Examination Survey 2003–2004". Environmental Health Perspectives. 116 (7): 893–7. doi:10.1289/ehp.11269. PMC 2453157. PMID 18629311.
- ↑ Gonzalez, H.; Farbrot, A.; Larko, O.; Wennberg, A. M. (2006). "Percutaneous absorption of the sunscreen benzophenone-3 after repeated whole-body applications, with and without ultraviolet irradiation". British Journal of Dermatology. 154 (2): 337–40. doi:10.1111/j.1365-2133.2005.07007.x. PMID 16433806.
- ↑ Hanson, K. M.; Gratton, E; Bardeen, C. J. (2006). "Sunscreen enhancement of UV-induced reactive oxygen species in the skin". Free Radical Biology and Medicine. 41 (8): 1205–12. doi:10.1016/j.freeradbiomed.2006.06.011. PMID 17015167.
- 1 2 3 Chisvert, A; León-González, Z; Tarazona, I; Salvador, A; Giokas, D (2012). "An overview of the analytical methods for the determination of organic ultraviolet filters in biological fluids and tissues". Analytica Chimica Acta. 752: 11–29. doi:10.1016/j.aca.2012.08.051. PMID 23101648.
- ↑ Okereke, C. S.; Kadry, A. M.; Abdel-Rahman, M. S.; Davis, R. A.; Friedman, M. A. (1993). "Metabolism of benzophenone-3 in rats". Drug Metabolism and Disposition. 21 (5): 788–91. PMID 7902237.
- ↑ Okereke, C. S.; Abdel-Rhaman, M. S.; Friedman, M. A. (1994). "Disposition of benzophenone-3 after dermal administration in male rats". Toxicology letters. 73 (2): 113–22. doi:10.1016/0378-4274(94)90101-5. PMID 8048080.
- 1 2 Sarveiya, V; Risk, S; Benson, H. A. (2004). "Liquid chromatographic assay for common sunscreen agents: Application to in vivo assessment of skin penetration and systemic absorption in human volunteers". Journal of Chromatography B. 803 (2): 225–31. doi:10.1016/j.jchromb.2003.12.022. PMID 15063329.
- ↑ "Hazardous Substances Data Bank". 2,4-Dihydroxybenzophenone. National Library of Medicine (US), Division of Specialized Information Services. Retrieved 19 April 2014.
- ↑ "Protect Yourself, Protect The Reef! The impacts of sunscreens on our coral reefs" (PDF). U.S. National Park Service. Retrieved 1 July 2013.
- ↑ Than, Ker. "Swimmers' Sunscreen Killing Off Coral". National Geographic News. National Geographic News. Retrieved January 29, 2008.
- ↑ Dowd, Katie (21 October 2015). "Study shows sunscreen is killing coral reefs in tourist areas". San Francisco Chronicle.
- ↑ "NICNAS Cosmetics Guidelines". Australian Government Department of Health. Archived from the original on 22 July 2013. Retrieved 9 March 2014.
- ↑ "Guidance Document Sunscreen Monograph". Health Canada. Retrieved 9 March 2014.
- ↑ Aguirre, Claudia. "Shedding Light on Sun Safety – Part Two". The International Dermal Institute. Retrieved 9 March 2014.
- ↑ http://eur-lex.europa.eu/legal-content/EN/ALL/?uri=uriserv:OJ.L_.2017.036.01.0037.01.ENG
- ↑ "Standards for Cosmetics" (PDF). Ministry of Health and Welfare Notification No.331 of 2000. Japanese Government. Retrieved 9 March 2014.
- ↑ "Suncreen Drug Products for Over-the-Counter Human Use". Code of Federal Regulations Title 21. FDA. Retrieved 9 March 2014.
- ↑ Folley, Aris (2 May 2018). "Hawaii lawmakers approve ban on sunscreens with chemicals harmful to coral reefs". The Hill. Retrieved 2 May 2018.
