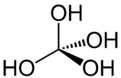
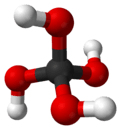
Orthocarbonic acid
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
orthocarbonic acid | |||
| Systematic IUPAC name
Methanetetrol[1] (substitutive) | |||
| Identifiers | |||
3D model (JSmol) |
|||
| ChemSpider | |||
PubChem CID |
|||
| |||
| |||
| Properties | |||
| CH4O4 | |||
| Molar mass | 80.04 g·mol−1 | ||
| Related compounds | |||
Related compounds |
Dihydroxymethylidene | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |||
| Infobox references | |||
Orthocarbonic acid (methanetetrol) is the name given to a hypothetical compound with the chemical formula H4CO4 or C(OH)4. Its molecular structure consists of a single carbon atom bonded to four hydroxy groups. It would be therefore a fourfold alcohol. In theory it could lose four protons to give the hypothetical oxocarbon anion CO4−
4 (orthocarbonate), and is therefore considered an oxoacid of carbon.
Orthocarbonic acid is highly unstable. Calculations show that it decomposes spontaneously into carbonic acid and H2O:[2][3]
- H4CO4 → H2CO3 + H2O.
Orthocarbonic acid is one of the group of carboxylic ortho acids that have the general structure of RC(OH)3.The term ortho acid is also used to refer to the most hydroxylated acid in a set of oxoacids. When drawn in two dimensions, the molecule resembles a swastika, except when we draw only hydroxy groups and the carbon is only as center, and has therefore been called "Hitler's acid".[4]
Researchers at the Moscow Institute of Physics and Technology predict that orthocarbonic acid is stable at high pressure, hence it may form in the interior of planets.[5][4]
Orthocarbonate anions
By loss of one through four protons, orthocarbonic acid could yield four anions: H
3CO−
4, H
2CO2−
4, HCO3−
4, and CO4−
4.
As of 2002, salts of these anions had yet to be observed. However, theoretical studies suggest that Na4CO4 might be stable.[6]
Orthocarbonate esters
The tetravalent moiety CO4 is found in stable organic compounds; they are formally esters of orthocarbonic acid, and therefore are called orthocarbonates. For example, Tetraethoxymethane can be prepared by the reaction between chloropicrin and sodium ethoxide in ethanol.[7] Polyorthocarbonates are stable polymers that might have applications in absorbing organic solvents in waste treatment processes,[8] or in dental restorative materials.[9] The explosive trinitroethylorthocarbonate possesses a orthocarbonate core.
See also
References
- ↑ "Methanetetrol - PubChem Public Chemical Database". The PubChem Project. USA: National Center for Biotechnology Information.
- ↑ Bohm S.; Antipova D.; Kuthan J. (1997). "A Study of Methanetetraol Dehydration to Carbonic Acid". International Journal of Quantum Chemistry. 62: 315–322. doi:10.1002/(SICI)1097-461X(1997)62:3<315::AID-QUA10>3.0.CO;2-8.
- ↑ Carboxylic Acids and Derivatives IUPAC Recommendations on Organic & Biochemical Nomenclature
- 1 2 GRAY, Richard (2016-09-07). "Weird materials like 'Hitler's Acid' hide inside Neptune and Uranus". Dailymail. Retrieved 2017-12-22.
- ↑ G. Saleh; A. R. Oganov (2016). "Novel Stable Compounds in the C-H-O Ternary System at High Pressure". Scientific Reports. doi:10.1038/srep32486.
- ↑ Al-Shemali Musstafa; Boldyre Alexander I (2002). "Search for Ionic Orthocarbonates: Ab Initio Study of Na4CO4". J. Phys. Chem. A. 106 (38): 8951–8954. doi:10.1021/jp020207+.
- ↑ Orthocarbonic acid, tetraethyl ester Organic Syntheses, Coll. Vol. 4, p.457 (1963); Vol. 32, p.68 (1952)
- ↑ Sonmez, H.B.; Wudl, F. (2005). "Cross-linked poly(orthocarbonate)s as organic solvent sorbents". Macromolecules. 38 (5): 1623–1626. doi:10.1021/ma048731x.
- ↑ Stansbury, J.W. (1992). "Synthesis and evaluation of new oxaspiro monomers for double ring-opening polymerization". Journal of Dental Research. 71 (7): 1408–1412. doi:10.1177/00220345920710070901. PMID 1629456. Archived from the original on 2008-07-08. Retrieved 2008-06-19.