Cortactin
Cortactin (from "cortical actin binding protein") is a monomeric protein located in the cytoplasm of cells that can be activated by external stimuli to promote polymerization and rearrangement of the actin cytoskeleton, especially the actin cortex around the cellular periphery.[5][6] It is present in all cell types. When activated, it will recruit Arp2/3 complex proteins to existing actin microfilaments, facilitating and stabilizing nucleation sites for actin branching. Cortactin is important in promoting lamellipodia formation, invadopodia formation, cell migration, and endocytosis.
Gene
In humans, cortactin is encoded by the CTTN gene on chromosome 11.[7]
Structure
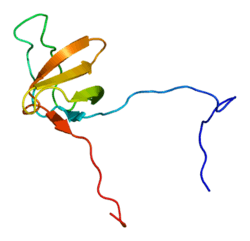
Cortactin is a thin, elongated monomer that consists of an amino-terminal acidic (NTA) region; 37-residue-long segments that are highly conserved among cortactin proteins of all species and repeated up to 6.5 times in tandem (“cortactin repeats”); a proline-rich region; and an SH3 domain. This basic structure is highly conserved among all species that express cortactin.[8]
Activation and binding
Cortactin is activated via phosphorylation, by tyrosine kinases or serine/threonine kinases, in response to extracellular signals like growth factors, adhesion sites, or pathogenic invasion of the epithelial layer.
The SH3 domain of certain tyrosine kinases, such as the oncogene Src kinase, binds to cortactin’s proline-rich region and phosphorylates it on Tyr421, Tyr466, and Tyr482. Once activated in this way, it can bind to filamentous actin (F-actin) with the fourth of its cortactin repeats.[8] As the concentration of phosphorylated cortactin increases in specific regions within the cell, the monomers each begin to recruit an Arp2/3 complex to F-actin. It binds to Arp2/3 with an aspartic acid-aspartic acid-tryptophan (DDW) sequence in its NTA region, a motif that is often seen in other actin nucleation-promoting factors (NPFs).[9]
Certain serine/threonine kinases, such as ERK, can phosphorylate cortactin on Ser405 and Ser418 in the SH3 domain.[8] Activated like this, it still associates with Arp2/3 and F-actin, but will also allow other actin NPFs, most importantly N-WASp (Neuronal Wiskott-Aldrich syndrome protein), to bind to the complex as well; when phosphorylated by tyrosine kinases, other NPFs are excluded.[10] The ability of these other NPFs to bind the Arp2/3 complex while cortactin is also bound could come from new interactions with cortactin’s SH3 domain, which is in a different conformation when phosphorylated by Ser/Thr kinases and thus may be more open to interactions with other NPFs.[10] Having other NPFs bind to the Arp2/3 complex at the same time as cortactin may enhance nucleation site stability.[8]
Location and function in the cell
Inactive cortactin diffuses throughout the cytoplasm, but upon phosphorylation, the protein begins to target certain areas in the cell. Cortactin-assisted Arp2/3-nucleated actin branches are most prominent in the actin cortex, around the periphery of the cell.[11] A phosphorylated cortactin monomer binds to, activates, and stabilizes an Arp2/3 complex on preexisting F-actin, which provides a nucleation site for a new actin branch to form from the “mother” filament. Branches formed from cortactin-assisted nucleation sites are very stable; cortactin has been shown to inhibit debranching.[11] Thus, polymerization and branching of actin is promoted in areas of the cell where cortactin is localized.
Cortactin is very active in lamellipodia, protrusions of the cell membrane formed by actin polymerization and treadmilling that propel the cell along a surface as it migrates towards some target.[12]
Cortactin acts as a link between extracellular signals and lamellipodial “steering.” When a receptor tyrosine kinase on the cell membrane binds to an adhesion site, for example, cortactin will be phosphorylated locally to the area of binding, activate and recruit Arp2/3 to the actin cortex in that region, and thus stimulate cortical actin polymerization and movement of the cell in that direction. Macrophages, highly motile immune cells that engulf cellular debris and pathogens, are propelled by lamellipodia and identify/migrate toward a target via chemotaxis; thus, cortactin must also be activated by receptor kinases that pick up a large variety of chemical signals.[12]
Studies have implicated cortactin in both clathrin-mediated endocytosis[13] and clathrin-independent endocytosis.[14] In both kinds of endocytosis, it has long been known that actin localizes to sites of vesicle invagination and is a vital part of the endocytic pathway, but the actual mechanisms by which actin facilitates endocytosis are still unclear. Recently, however, it has been found that dynamin, the protein responsible for breaking the newly formed vesicular bud off the inside of the plasma membrane, can associate with the SH3 domain of cortactin. Since cortactin recruits the Arp2/3 complexes that lead to actin polymerization, this suggests that it may play an important part in linking vesicle formation to the as yet unknown functions actin has in endocytosis.[15]
Clinical significance
Amplification of the genes encoding cortactin—in humans, EMS1—has been found to occur in certain tumors. Overexpression of cortactin can lead to highly-active lamellipodia in tumor cells, dubbed “invadopodia.” These cells are especially invasive and migratory, making them very dangerous, for they can easily spread cancer across the body into other tissues.[16]
Interactions
Cortactin has been shown to interact with:
See also
References
- 1 2 3 GRCh38: Ensembl release 89: ENSG00000085733 - Ensembl, May 2017
- 1 2 3 GRCm38: Ensembl release 89: ENSMUSG00000031078 - Ensembl, May 2017
- ↑ "Human PubMed Reference:".
- ↑ "Mouse PubMed Reference:".
- ↑ Cosen-Binker LI, Kapus A (October 2006). "Cortactin: the gray eminence of the cytoskeleton". Physiology. 21 (5): 352–61. doi:10.1152/physiol.00012.2006. PMID 16990456.
- ↑ Ammer AG, Weed SA (September 2008). "Cortactin branches out: roles in regulating protrusive actin dynamics". Cell Motil. Cytoskeleton. 65 (9): 687–707. doi:10.1002/cm.20296. PMC 2561250. PMID 18615630.
- ↑ Brookes S, Lammie GA, Schuuring E, de Boer C, Michalides R, Dickson C, Peters G (April 1993). "Amplified region of chromosome band 11q13 in breast and squamous cell carcinomas encompasses three CpG islands telomeric of FGF3, including the expressed gene EMS1". Genes Chromosomes Cancer. 6 (4): 222–31. doi:10.1002/gcc.2870060406. PMID 7685625.
- 1 2 3 4 Daly RJ (August 2004). "Cortactin signalling and dynamic actin networks". Biochem. J. 382 (Pt 1): 13–25. doi:10.1042/BJ20040737. PMC 1133910. PMID 15186216.
- ↑ Weed SA, Karginov AV, Schafer DA, Weaver AM, Kinley AW, Cooper JA, Parsons JT (October 2000). "Cortactin localization to sites of actin assembly in lamellipodia requires interactions with F-actin and the Arp2/3 complex". J. Cell Biol. 151 (1): 29–40. doi:10.1083/jcb.151.1.29. PMC 2189811. PMID 11018051.
- 1 2 Martinez-Quiles N, Ho HY, Kirschner MW, Ramesh N, Geha RS (June 2004). "Erk/Src phosphorylation of cortactin acts as a switch on-switch off mechanism that controls its ability to activate N-WASP". Mol. Cell. Biol. 24 (12): 5269–80. doi:10.1128/MCB.24.12.5269-5280.2004. PMC 419870. PMID 15169891.
- 1 2 Weaver AM, Karginov AV, Kinley AW, Weed SA, Li Y, Parsons JT, Cooper JA (March 2001). "Cortactin promotes and stabilizes Arp2/3-induced actin filament network formation". Curr. Biol. 11 (5): 370–4. doi:10.1016/S0960-9822(01)00098-7. PMID 11267876.
- 1 2 Weed SA, Parsons JT (October 2001). "Cortactin: coupling membrane dynamics to cortical actin assembly". Oncogene. 20 (44): 6418–34. doi:10.1038/sj.onc.1204783. PMID 11607842.
- ↑ Samaj J, Baluska F, Voigt B, Schlicht M, Volkmann D, Menzel D (July 2004). "Endocytosis, actin cytoskeleton, and signaling". Plant Physiol. 135 (3): 1150–61. doi:10.1104/pp.104.040683. PMC 519036. PMID 15266049.
- ↑ Sauvonnet N, Dujeancourt A, Dautry-Varsat A (January 2005). "Cortactin and dynamin are required for the clathrin-independent endocytosis of gammac cytokine receptor". J. Cell Biol. 168 (1): 155–63. doi:10.1083/jcb.200406174. PMC 2171671. PMID 15623579.
- ↑ Zhu J, Zhou K, Hao JJ, Liu J, Smith N, Zhan X (February 2005). "Regulation of cortactin/dynamin interaction by actin polymerization during the fission of clathrin-coated pits". J. Cell Sci. 118 (Pt 4): 807–17. doi:10.1242/jcs.01668. PMID 15671060.
- ↑ Weaver AM (2006). "Invadopodia: specialized cell structures for cancer invasion". Clin. Exp. Metastasis. 23 (2): 97–105. doi:10.1007/s10585-006-9014-1. PMID 16830222.
- 1 2 Weed SA, Karginov AV, Schafer DA, Weaver AM, Kinley AW, Cooper JA, Parsons JT (October 2000). "Cortactin localization to sites of actin assembly in lamellipodia requires interactions with F-actin and the Arp2/3 complex". J. Cell Biol. 151 (1): 29–40. doi:10.1083/jcb.151.1.29. PMC 2189811. PMID 11018051.
- ↑ Di Ciano C, Nie Z, Szászi K, Lewis A, Uruno T, Zhan X, Rotstein OD, Mak A, Kapus A (September 2002). "Osmotic stress-induced remodeling of the cortical cytoskeleton". Am. J. Physiol., Cell Physiol. 283 (3): C850–65. doi:10.1152/ajpcell.00018.2002. PMID 12176742.
- ↑ Martinez MC, Ochiishi T, Majewski M, Kosik KS (July 2003). "Dual regulation of neuronal morphogenesis by a delta-catenin-cortactin complex and Rho". J. Cell Biol. 162 (1): 99–111. doi:10.1083/jcb.200211025. PMC 2172717. PMID 12835311.
- ↑ Kim L, Wong TW (September 1998). "Growth factor-dependent phosphorylation of the actin-binding protein cortactin is mediated by the cytoplasmic tyrosine kinase FER". J. Biol. Chem. 273 (36): 23542–8. doi:10.1074/jbc.273.36.23542. PMID 9722593.
- ↑ Hattan D, Nesti E, Cachero TG, Morielli AD (October 2002). "Tyrosine phosphorylation of Kv1.2 modulates its interaction with the actin-binding protein cortactin". J. Biol. Chem. 277 (41): 38596–606. doi:10.1074/jbc.M205005200. PMID 12151401.
- ↑ Du Y, Weed SA, Xiong WC, Marshall TD, Parsons JT (October 1998). "Identification of a novel cortactin SH3 domain-binding protein and its localization to growth cones of cultured neurons". Mol. Cell. Biol. 18 (10): 5838–51. PMC 109170. PMID 9742101.
- ↑ Mizutani K, Miki H, He H, Maruta H, Takenawa T (February 2002). "Essential role of neural Wiskott-Aldrich syndrome protein in podosome formation and degradation of extracellular matrix in src-transformed fibroblasts". Cancer Res. 62 (3): 669–74. PMID 11830518.
- ↑ Kinley AW, Weed SA, Weaver AM, Karginov AV, Bissonette E, Cooper JA, Parsons JT (March 2003). "Cortactin interacts with WIP in regulating Arp2/3 activation and membrane protrusion". Curr. Biol. 13 (5): 384–93. doi:10.1016/s0960-9822(03)00107-6. PMID 12620186.
Further reading
- Weed SA, Parsons JT (2001). "Cortactin: coupling membrane dynamics to cortical actin assembly". Oncogene. 20 (44): 6418–34. doi:10.1038/sj.onc.1204783. PMID 11607842.
- Buday L, Downward J (2007). "Roles of cortactin in tumor pathogenesis". Biochim. Biophys. Acta. 1775 (2): 263–73. doi:10.1016/j.bbcan.2006.12.002. PMID 17292556.
- Schuuring E, Verhoeven E, Mooi WJ, Michalides RJ (1992). "Identification and cloning of two overexpressed genes, U21B31/PRAD1 and EMS1, within the amplified chromosome 11q13 region in human carcinomas". Oncogene. 7 (2): 355–61. PMID 1532244.
- Wu H, Parsons JT (1993). "Cortactin, an 80/85-kilodalton pp60src substrate, is a filamentous actin-binding protein enriched in the cell cortex". J. Cell Biol. 120 (6): 1417–26. doi:10.1083/jcb.120.6.1417. PMC 2119758. PMID 7680654.
- Brookes S, Lammie GA, Schuuring E, de Boer C, Michalides R, Dickson C, Peters G (1993). "Amplified region of chromosome band 11q13 in breast and squamous cell carcinomas encompasses three CpG islands telomeric of FGF3, including the expressed gene EMS1". Genes Chromosomes Cancer. 6 (4): 222–31. doi:10.1002/gcc.2870060406. PMID 7685625.
- Maruyama K, Sugano S (1994). "Oligo-capping: a simple method to replace the cap structure of eukaryotic mRNAs with oligoribonucleotides". Gene. 138 (1–2): 171–4. doi:10.1016/0378-1119(94)90802-8. PMID 8125298.
- Schuuring E, Verhoeven E, Litvinov S, Michalides RJ (1993). "The product of the EMS1 gene, amplified and overexpressed in human carcinomas, is homologous to a v-src substrate and is located in cell-substratum contact sites". Mol. Cell. Biol. 13 (5): 2891–98. PMC 359682. PMID 8474448.
- Maruyama S, Kurosaki T, Sada K, Yamanashi Y, Yamamoto T, Yamamura H (1996). "Physical and functional association of cortactin with Syk in human leukemic cell line K562". J. Biol. Chem. 271 (12): 6631–5. doi:10.1074/jbc.271.26.15615. PMID 8636079.
- van Damme H, Brok H, Schuuring-Scholtes E, Schuuring E (1997). "The redistribution of cortactin into cell-matrix contact sites in human carcinoma cells with 11q13 amplification is associated with both overexpression and post-translational modification". J. Biol. Chem. 272 (11): 7374–80. doi:10.1074/jbc.272.11.7374. PMID 9054437.
- Suzuki Y, Yoshitomo-Nakagawa K, Maruyama K, Suyama A, Sugano S (1997). "Construction and characterization of a full length-enriched and a 5'-end-enriched cDNA library". Gene. 200 (1–2): 149–56. doi:10.1016/S0378-1119(97)00411-3. PMID 9373149.
- Kinnunen T, Kaksonen M, Saarinen J, Kalkkinen N, Peng HB, Rauvala H (1998). "Cortactin-Src kinase signaling pathway is involved in N-syndecan-dependent neurite outgrowth". J. Biol. Chem. 273 (17): 10702–8. doi:10.1074/jbc.273.17.10702. PMID 9553134.
- Kim L, Wong TW (1998). "Growth factor-dependent phosphorylation of the actin-binding protein cortactin is mediated by the cytoplasmic tyrosine kinase FER". J. Biol. Chem. 273 (36): 23542–8. doi:10.1074/jbc.273.36.23542. PMID 9722593.
- Du Y, Weed SA, Xiong WC, Marshall TD, Parsons JT (1998). "Identification of a novel cortactin SH3 domain-binding protein and its localization to growth cones of cultured neurons". Mol. Cell. Biol. 18 (10): 5838–51. PMC 109170. PMID 9742101.
- Huang C, Liu J, Haudenschild CC, Zhan X (1998). "The role of tyrosine phosphorylation of cortactin in the locomotion of endothelial cells". J. Biol. Chem. 273 (40): 25770–6. doi:10.1074/jbc.273.40.25770. PMID 9748248.
- Katsube T, Takahisa M, Ueda R, Hashimoto N, Kobayashi M, Togashi S (1998). "Cortactin associates with the cell-cell junction protein ZO-1 in both Drosophila and mouse". J. Biol. Chem. 273 (45): 29672–7. doi:10.1074/jbc.273.45.29672. PMID 9792678.
- Ohoka Y, Takai Y (1998). "Isolation and characterization of cortactin isoforms and a novel cortactin-binding protein, CBP90". Genes Cells. 3 (9): 603–12. doi:10.1046/j.1365-2443.1998.00216.x. PMID 9813110.
- Schuuring E, van Damme H, Schuuring-Scholtes E, Verhoeven E, Michalides R, Geelen E, de Boer C, Brok H, van Buuren V, Kluin P (1999). "Characterization of the EMS1 gene and its product, human Cortactin". Cell Adhes. Commun. 6 (2–3): 185–209. doi:10.3109/15419069809004475. PMID 9823470.
- Campbell DH, Sutherland RL, Daly RJ (1999). "Signaling pathways and structural domains required for phosphorylation of EMS1/cortactin". Cancer Res. 59 (20): 5376–85. PMID 10537323.
- Kapus A, Di Ciano C, Sun J, Zhan X, Kim L, Wong TW, Rotstein OD (2000). "Cell volume-dependent phosphorylation of proteins of the cortical cytoskeleton and cell-cell contact sites. The role of Fyn and FER kinases". J. Biol. Chem. 275 (41): 32289–98. doi:10.1074/jbc.M003172200. PMID 10921917.
- Weed SA, Karginov AV, Schafer DA, Weaver AM, Kinley AW, Cooper JA, Parsons JT (2000). "Cortactin localization to sites of actin assembly in lamellipodia requires interactions with F-actin and the Arp2/3 complex". J. Cell Biol. 151 (1): 29–40. doi:10.1083/jcb.151.1.29. PMC 2189811. PMID 11018051.
External links
- Cortactin at the US National Library of Medicine Medical Subject Headings (MeSH)