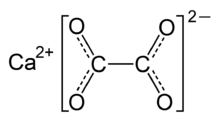
Calcium oxalate
 | |
| Identifiers | |
|---|---|
| |
3D model (JSmol) |
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.008.419 |
PubChem CID |
|
| |
| |
| Properties | |
| C2H2CaO5 | |
| Molar mass | 146.11 g·mol−1 |
| Appearance | white solid |
| Density | 2.20 g/cm3, monohydrate[1] |
| Melting point | 200 °C (392 °F; 473 K) decomposes (monohydrate) |
| 0.67 mg/L (20 °C) | |
| Related compounds | |
Other cations |
Beryllium oxalate Magnesium oxalate Strontium oxalate Barium oxalate Radium oxalate Iron(II) oxalate Iron(III) oxalate |
Related compounds |
Oxalic acid |
| Hazards | |
| Main hazards | Harmful |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |


Calcium oxalate (in archaic terminology, oxalate of lime) is a calcium salt of oxalate with the chemical formula CaC2O4(H2O)x, where x can vary. All forms are colorless or white. The monohydrate occurs naturally as the mineral whewellite, forming envelope-shaped crystals, known in plants as raphides. The rarer dihydrate (mineral: weddellite) and trihydrate (mineral: caoxite) are also recognized. Calcium oxalates are a major constituent of human kidney stones, calcium oxalate is also found in beerstone, a scale that forms on containers used in breweries.
Occurrence
Many plants accumulate calcium oxalate as it has been reported in more than 1000 different genera of plants.[2] The calcium oxalate accumulation is linked to the detoxification of calcium (Ca2+) in the plant.[3]
The poisonous plant dumb cane (Dieffenbachia) contains the substance and on ingestion can prevent speech and be suffocating. It is also found in sorrel, rhubarb (in large quantities in the leaves), cinnamon, turmeric and in species of Oxalis, Araceae, Arum Italicum, taro, kiwifruit, tea leaves, agaves, Virginia creeper (Parthenocissus quinquefolia), and Alocasia and in spinach in varying amounts. Plants of the genus Philodendron contain enough calcium oxalate that consumption of parts of the plant can result in uncomfortable symptoms. Insoluble calcium oxalate crystals are found in plant stems, roots, and leaves and produced in idioblasts.
Calcium oxalate, as ‘beerstone’, is a brownish precipitate that tends to accumulate within vats, barrels, and other containers used in the brewing of beer. If not removed in a cleaning process, beerstone will leave an unsanitary surface that can harbour microorganisms.[4] Beerstone is composed of calcium and magnesium salts and various organic compounds left over from the brewing process; it promotes the growth of unwanted microorganisms that can adversely affect or even ruin the flavour of a batch of beer.
Calcium oxalate crystals in the urine are the most common constituent of human kidney stones, and calcium oxalate crystal formation is also one of the toxic effects of ethylene glycol poisoning.
Medical significance
Calcium oxalate can produce sores and numbing on ingestion and may even be fatal.
Morphology and diagnosis
The monohydrate and dihydrate can be distinguished by the shape of the respective crystals.
- Calcium oxalate dihydrate crystals are octahedral. A large portion of the crystals in a urine sediment will have this type of morphology, as they can grow at any pH and naturally occur in normal urine.
- Calcium oxalate monohydrate crystals vary in shape, and can be shaped like dumbbells, spindles, ovals, or picket fences, the last of which is most commonly seen due to ethylene glycol poisoning.[5] This latter form is what investigators discovered in the kidneys of two victims that ultimately led to the successful prosecution and conviction of murderer Lynn Turner, who poisoned both her husband and boyfriend with ethylene glycol-based antifreeze.[6]
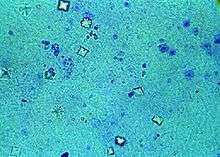 Urine microscopy showing calcium oxalate crystals in the urine. The octahedral crystal morphology is clearly visible.
Urine microscopy showing calcium oxalate crystals in the urine. The octahedral crystal morphology is clearly visible._-_kalsiyum_oksalat_kristalleri_(idrar)_-_01.png) Urine microscopy showing a calcium oxalate monohydrate crystal (dumbbell shaped) and a calcium oxalate dihydrate crystal (envelope shaped) along with several erythrocytes.
Urine microscopy showing a calcium oxalate monohydrate crystal (dumbbell shaped) and a calcium oxalate dihydrate crystal (envelope shaped) along with several erythrocytes._-_kalsiyum_oksalat_kristalleri_(idrar)_-_02.png) Urine microscopy showing several calcium oxalate monohydrate crystals (dumbbell shaped, some of them clumped) and a calcium oxalate dihydrate crystal (envelope shaped) along with several erythrocytes.
Urine microscopy showing several calcium oxalate monohydrate crystals (dumbbell shaped, some of them clumped) and a calcium oxalate dihydrate crystal (envelope shaped) along with several erythrocytes.
Kidney stones
About 80% of kidney stones are partially or entirely of the calcium oxalate type. They form when urine is persistently saturated with calcium and oxalate. Some of the oxalate in urine is produced by the body. Calcium and oxalate in the diet play a part, but are not the only factors that affect the formation of calcium oxalate stones. Dietary oxalate is an organic ion found in many vegetables, fruits, and nuts. Calcium from bone may also play a role in kidney stone formation.
Industrial applications
Calcium oxalate is used in the manufacture of ceramic glazes.[7]
References
- 1 2 S. Deganello (1981). "The Structure of Whewellite, CaC204.H20 , at 328 K". Acta Crystallogr. B. 37: 826–829. doi:10.1107/S056774088100441X.
- ↑ Francesci, V.R.; Nakata (2005). "Calcium oxalate in plants: formation and function". Annu Rev Plant Biol (56): 41–71.
- ↑ Martin, G; Matteo Guggiari; Daniel Bravo; Jakob Zopfi; Guillaume Cailleau; Michel Aragno; Daniel Job; Eric Verrecchia; Pilar Junier (2012). "Fungi, bacteria and soil pH: the oxalate–carbonate pathway as a model for metabolic interaction". Environmental Microbiology. 14 (11): 2960–2970. doi:10.1111/j.1462-2920.2012.02862.x. PMID 22928486.
- ↑ Ryan, James (27 May 2018). "What is beerstone (and how to remove it)". Retrieved 2018-05-28.
- ↑ "Urine Crystals". ahdc.vet.cornell.edu/. Cornell University. Retrieved 12 July 2014.
- ↑ Nash, Alanna. "The Black Widow Killer: Two men. Two murders. Too many questions". Reader's Digest. Retrieved 2009-04-26.
- ↑ "Calcium Oxalate Data Sheet". Hummel Croton Inc. Retrieved 2017-04-23.