4-Vinylpyridine
 | |
| Names | |
|---|---|
| IUPAC name
4-ethenylpyridine | |
| Identifiers | |
3D model (JSmol) |
|
| 104506 | |
| ChemSpider | |
| ECHA InfoCard | 100.002.593 |
| EC Number | 202-852-0 |
PubChem CID |
|
| UNII | |
| |
| |
| Properties | |
| C7H7N | |
| Molar mass | 105.14 g·mol−1 |
| Appearance | colorless liquid |
| Density | 0.988 g/cm3 |
| Boiling point | 62–65 °C (144–149 °F; 335–338 K) 15 mmHg |
| Hazards | |
| GHS pictograms |  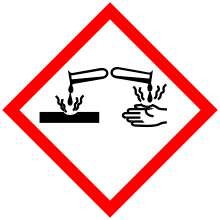     |
| GHS signal word | Danger |
| H226, H301, H314, H315, H317, H319, H330, H334, H411 | |
| P210, P233, P240, P241, P242, P243, P260, P261, P264, P270, P271, P272, P273, P280, P284, P285, P301+310, P301+330+331, P302+352, P303+361+353, P304+340, P304+341, P305+351+338, P310, P320 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
4-Vinylpyridine is an organic compound with the formula CH2CHC5H4N. It is a derivative of pyridine with a vinyl group in the 4-position. It is a colorless liquid, although impure samples are often brown. It is a monomeric precursor to specialty polymers. 4-Vinylpyridine is prepared by the condensation of 4-methylpyridine and formaldehyde.[1]
See also
References
- ↑ Abe, Nobuyuki; Ichimura, Hisao; Kataoka, Toshiaki; Morishita, Sinji; Shimizu, Shinkichi; Shoji, Takayuki; Watanabe, Nanao. (2007). Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a22_399.
This article is issued from
Wikipedia.
The text is licensed under Creative Commons - Attribution - Sharealike.
Additional terms may apply for the media files.