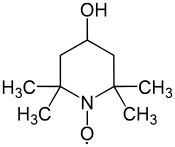
4-Hydroxy-TEMPO
 | |
 | |
| Names | |
|---|---|
| IUPAC name
1-λ1-oxidanyl-2,2,6,6-tetramethylpiperidin-4-ol | |
| Other names
tempol; tanol; TMPN; 4-Oxypiperidol; nitroxyl 2; HyTEMPO | |
| Identifiers | |
3D model (JSmol) |
|
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.017.056 |
PubChem CID |
|
| |
| |
| Properties | |
| C9H18NO2 | |
| Molar mass | 172.25 g·mol−1 |
| Appearance | Orange crystals |
| Melting point | 71–73 °C (160–163 °F; 344–346 K)[1] |
| Hazards | |
| GHS pictograms |  |
| GHS signal word | Warning[2] |
| H302, H315, H319, H335[2] | |
| P261, P305+351+338[2] | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
4-Hydroxy-TEMPO or TEMPOL, formally 4-hydroxy-2,2,6,6-tetramethylpiperidin-1-oxyl, is a heterocyclic compound. Like the related TEMPO, it is used as a catalyst and chemical oxidant by virtue of being a stable radical. Its major appeal over TEMPO is that is less expensive, being produced from triacetone amine, which is itself made via the condensation of acetone and ammonia. This makes it economically viable on an industrial scale.[3]
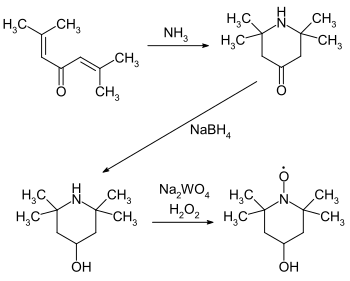
In biochemical research, it has been investigated as an agent for limiting reactive oxygen species. It catalyzes the disproportionation of superoxide, facilitates hydrogen peroxide metabolism, and inhibits Fenton chemistry.[4] It is often present as a structural element in hindered amine light stabilizers, which are commonly used stabilizers in plastics.
References
- ↑ Zakrzewski, Jerzy; Krawczyk, Maria (1 January 2011). "Reactions of Nitroxides. Part XII [1]. – 2,2,6,6-Tetramethyl-1-oxyl- 4-piperidyl Chloroformate – A New Reactive Nitroxyl Radical. A One-pot Synthesis of 2,2,6,6-Tetramethyl-1-oxyl-4-piperidyl N,N-Dialkyl-carbamates". Zeitschrift für Naturforschung B. 66 (5). doi:10.1515/znb-2011-0509.
- 1 2 3 4 Sigma-Aldrich Co., 4-Hydroxy-TEMPO. Retrieved on 2015-08-24.
- ↑ Ciriminna, Rosaria; Pagliaro, Mario (15 January 2010). "Industrial Oxidations with Organocatalyst TEMPO and Its Derivatives". Organic Process Research & Development. 14 (1): 245–251. doi:10.1021/op900059x.
- ↑ Wilcox, C. S.; Pearlman, A. (2008). "Chemistry and Antihypertensive Effects of Tempol and Other Nitroxides". Pharmacological Reviews. 60 (4): 418–69. doi:10.1124/pr.108.000240. PMC 2739999. PMID 19112152.