Stibnite
Stibnite, sometimes called antimonite, is a sulfide mineral with the formula Sb2S3. This soft grey material crystallizes in an orthorhombic space group. It is the most important source for the metalloid antimony.[4] The name is from the Greek στίβι stibi through the Latin stibium as the old name for the mineral and the element antimony.[1][2]
| Stibnite | |
|---|---|
 | |
| General | |
| Category | Sulfide mineral |
| Formula (repeating unit) | Sb2S3 |
| Strunz classification | 2.DB.05a |
| Crystal system | Orthorhombic |
| Crystal class | Dipyramidal (mmm) H-M symbol: (2/m 2/m 2/m) |
| Space group | Pbnm |
| Unit cell | a = 11.229 Å, b = 11.31 Å, c = 3.8389 Å; Z = 4 |
| Identification | |
| Color | Lead-gray, tarnishing blackish or iridescent; in polished section, white |
| Crystal habit | Massive, radiating and elongated crystals. Massive and granular |
| Twinning | Rare |
| Cleavage | Perfect and easy on {010}; imperfect on {100} and {110} |
| Fracture | Subconchoidal |
| Tenacity | Highly flexible but not elastic; slightly sectile |
| Mohs scale hardness | 2 |
| Luster | Splendent on fresh crystals surfaces, otherwise metallic |
| Streak | Similar to color |
| Diaphaneity | Opaque |
| Specific gravity | 4.63 |
| Solubility | decomposed with hydrochloric acid |
| Other characteristics | Anisotropism: Strong |
| References | [1][2][3] |
| Major varieties | |
| Metastibnite | Earthy, reddish deposits |
Structure
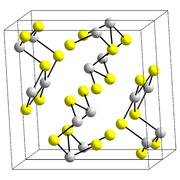
Stibnite has a structure similar to that of arsenic trisulfide, As2S3. The Sb(III) centers, which are pyramidal and three-coordinate, are linked via bent two-coordinate sulfide ions. However, recent studies confirm that the actual coordination polyhedra of antimony are in fact SbS7, with (3+4) coordination at the M1 site and (5+2) at the M2 site. Some of the secondary bonds impart cohesion and are connected with packing.[5] Stibnite is grey when fresh, but can turn superficially black due to oxidation in air.
Properties
The melting point of Sb2S3 is 823 K.[6] The band gap is 1.88 eV at room temperature and it is a photoconductor.[7]
Uses
.jpg)
Pastes of Sb2S3 powder in fat[8] or in other materials have been used since ca. 3000 BC as eye cosmetics in the Mediterranean and farther afield; in this use, Sb2S3 is called kohl. It was used to darken the brows and lashes, or to draw a line around the perimeter of the eye.
Antimony trisulfide finds use in pyrotechnic compositions, namely in the glitter and fountain mixtures. Needle-like crystals, "Chinese Needle", are used in glitter compositions and white pyrotechnic stars. The "Dark Pyro" version is used in flash powders to increase their sensitivity and sharpen their report. It is also a component of modern safety matches. It was formerly used in flash compositions, but its use was abandoned due to toxicity and sensitivity to static electricity.[9]
Stibnite was used ever since protodynastic Ancient Egypt as a medication and a cosmetic. The Sunan Abi Dawood reports, “prophet Muhammad said: 'Among the best types of collyrium is antimony (ithmid) for it clears the vision and makes the hair sprout.'”[10]
The 17th century alchemist Eirenaeus Philalethes, also known as George Starkey, describes stibnite in his alchemical commentary An Exposition upon Sir George Ripley's Epistle. Starkey used stibnite as a precursor to philosophical mercury, which was itself a hypothetical precursor to the Philosopher's stone.[11]
Occurrence
Stibnite occurs in hydrothermal deposits and is associated with realgar, orpiment, cinnabar, galena, pyrite, marcasite, arsenopyrite, cervantite, stibiconite, calcite, ankerite, barite and chalcedony.[1]
Small deposits of stibnite are common, but large deposits are rare. It occurs in Canada, Mexico, Peru, Japan, China, Germany, Romania, Italy, France, England, Algeria, and Kalimantan, Borneo. In the United States it is found in Arkansas, Idaho, Nevada, California, and Alaska.
As of May 2007, the largest specimen on public display (1000 pounds) is at the American Museum of Natural History.[12][13] The largest documented single crystals of stibnite measured ~60×5×5 cm and originated from different locations including Japan, France and Germany.[14]
 Sample from in the Carnegie Museum of Natural History
Sample from in the Carnegie Museum of Natural History Needles of stibnite within a transparent crystal of calcite (size: 4.5×3.5×1.8 cm)
Needles of stibnite within a transparent crystal of calcite (size: 4.5×3.5×1.8 cm) Ray of sharp, striated, iridescent metallic stibnite blades
Ray of sharp, striated, iridescent metallic stibnite blades
See also
References
- Stibnite. Handbook of Mineralogy
- Stibnite. Mindat.org
- Stibnite. Webmineral
- Sabina C. Grund, K. Hanusch, H. J. Breunig, H. U. Wolf, "Antimony and Antimony Compounds" in Ullmann’s Encyclopedia of Industrial Chemistry 2006, Wiley-VCH, Weinheim. doi:10.1002/14356007.a03_055.pub2
- Kyono A (2002). "Low-temperature crystal structures of stibnite implying orbital overlap of Sb 5s 2 inert pair electrons". Physics and Chemistry of Minerals. 29 (4): 254–260. Bibcode:2002PCM....29..254K. doi:10.1007/s00269-001-0227-1.
- Mills, K. C. (1974). Thermodynamic data for inorganic sulphides, selenides and tellurides. London: Butterworths. ISBN 040870537X. OCLC 1031663.
- Madelung, O. (Otfried) (2004). Semiconductors : data handbook (3rd ed.). Berlin: Springer. ISBN 3540404880. OCLC 56192440.
- Priesner, Claus; Figala, Karin, eds. (1998). Alchemie. Lexikon einer hermetischen Wissenschaft (in German). München: C.H. Beck. ISBN 978-3-406-44106-6.
- Pyrotechnic Chemical Guide. PyroUniverse.com. Retrieved on 2011-10-14.
- Sunan Abu-Dawud (Ahmad Hasan translation). Book 32, Number 4050.
- Eirenaeus Philalethes and Carl Jung http://www.persee.fr/web/revues/home/prescript/article/rhs_0151-4105_1996_num_49_2_1254
- "American Museum of Natural History, Spectacular Stibnite". American Museum of Natural History. Retrieved 2007-05-27.
- "Chinese stibnite crystal on display in US". Retrieved 2009-06-06.
- P. C. Rickwood (1981). "The largest crystals" (PDF). American Mineralogist. 66: 885–907.
External links
| Wikimedia Commons has media related to Stibnite. |
- Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica (11th ed.). Cambridge University Press.