Mobile genetic elements
Mobile genetic elements (MGEs) are a type of genetic material that can move around within a genome, or that can be transferred from one species or replicon to another. MGEs are found in all organisms. In humans, approximately 50% of the genome is thought to be MGEs.[1] MGEs play a distinct role in evolution. Gene duplication events can also happen through the mechanism of MGEs. MGEs can also cause mutations in protein coding regions, which alters the protein functions. They can also rearrange genes in the host genome. One of the examples of MGEs in evolutionary context is that virulence factors and antibiotic resistance genes of MGEs can be transported to share them with neighboring bacteria. Newly acquired genes through this mechanism can increase fitness by gaining new or additional functions. On the other hand, MGEs can also decrease fitness by introducing disease-causing alleles or mutations.[2]
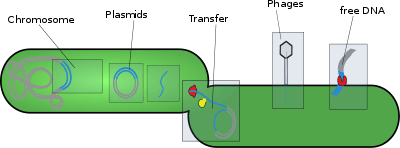
Types
- Transposons (also called transposable elements) are DNA sequences that can move locations within a genome, which includes retrotransposons and DNA transposons.
- Retrotransposons are the most widespread class of transposons in mammals.[3] An RNA transcript of MGEs is copied by reverse transcriptase. Then, the DNA sequence can be inserted back to a random location of the genome.[4]
- DNA transposons are a DNA segment that can move to a new location by a "cut-and-paste" strategy.
- Plasmids of bacteria are a transferable genetic element through bacterial conjugation. This is a mechanism of horizontal gene transfer that allows bacteria to share virulence factors and antibiotic resistance genes.
- Bacteriophage elements, like Mu, which integrates randomly into the genome by transduction.[5]
- Group I and Group II introns are a product from self-splicing in the host transcripts, and they act as ribozymes that can invade tRNA, rRNA, and protein coding genes in bacteria.[6]
Research examples
CRISPR-Cas systems in bacteria and archaea are adaptive immune systems to protect against deadly consequences from MGEs. Using comparative genomic and phylogenetic analysis, researchers found that CRISPR-Cas variants are associated with distinct types of MGEs such as transposable elements. In addition, CRISPR-Cas controls transposable elements for their propagation.[7]
MGEs such as plasmids by a horizontal transmission are generally beneficial to an organism. The ability of transferring plasmids (sharing) is important in an evolutionary perspective. Tazzyman and Bonhoeffer found that fixation (receiving) of the transferred plasmids in a new organism is just as important as the ability to transfer them.[8] Beneficial rare and transferable plasmids have a higher fixation probability, whereas deleterious transferable genetic elements have a lower fixation probability to avoid lethality to the host organisms.
Transposition by transposable elements is mutagenic. Thus, organisms have evolved to repress the transposition events, and failure to repress the events causes cancers in somatic cells. Cecco et al. found that during early age transcription of retrotransposable elements are minimal in mice, but in advanced age the transcription level increases.[9] This age-dependent expression level of transposable elements is reduced by calorie restriction diet.
Diseases
The consequence of mobile genetic elements can alter the transcriptional patterns, which frequently leads to genetic disorders such as immune disorders, breast cancer, multiple sclerosis, and amyotrophic lateral sclerosis. In humans, stress can lead to transactional activation of MGEs such as endogenous retroviruses, and this activation has been linked to neurodegeneration.[10]
Other notes
The total of all mobile genetic elements in a genome may be referred to as the mobilome.
Barbara McClintock was awarded the 1983 Nobel Prize in Physiology or Medicine "for her discovery of mobile genetic elements" (transposable elements).[11]
Mobile genetic elements play a critical role in the spread of virulence factors, such as exotoxins and exoenzymes, among bacteria. Strategies to combat certain bacterial infections by targeting these specific virulence factors and mobile genetic elements have been proposed.[12]
See also
- ACLAME (The CLAssification of Mobile genetic Elements) database
- Horizontal gene transfer
- Virulence factors
References
- Mu, X.; Ahmad, S.; Hur, S. (2016). Endogenous Retroelements and the Host Innate Immune Sensors. Advances in Immunology. 132. pp. 47–69. doi:10.1016/bs.ai.2016.07.001. ISBN 9780128047972. PMC 5135014. PMID 27769507.
- Singh, Parmit Kumar; Bourque, Guillaume; Craig, Nancy L.; Dubnau, Josh T.; Feschotte, Cédric; Flasch, Diane A.; Gunderson, Kevin L.; Malik, Harmit Singh; Moran, John V. (2014-11-18). "Mobile genetic elements and genome evolution 2014". Mobile DNA. 5: 26. doi:10.1186/1759-8753-5-26. PMC 4363357. PMID 30117500.
- Richardson, Sandra R.; Garcia-Perez, José Luis; Doucet, Aurélien J.; Kopera, Huira C.; Moldovan, John B.; Moran, John V. (2015-03-05). "The Influence of LINE-1 and SINE Retrotransposons on Mammalian Genomes". Microbiology Spectrum. 3 (2): 1165–1208. doi:10.1128/microbiolspec.mdna3-0061-2014. ISBN 9781555819200. PMC 4498412. PMID 26104698.
- Hsu, Ellen; Lewis, Susanna M. (2015). "The Origin of V(D)J Diversification". Molecular Biology of B Cells. pp. 133–149. doi:10.1016/b978-0-12-397933-9.00009-6. ISBN 9780123979339.
- Rankin, D. J.; Rocha, E. P. C.; Brown, S. P. (January 2011). "What traits are carried on mobile genetic elements, and why?". Heredity. 106 (1): 1–10. doi:10.1038/hdy.2010.24. PMC 3183850. PMID 20332804.
- Hausner, Georg; Hafez, Mohamed; Edgell, David R. (2014-03-10). "Bacterial group I introns: mobile RNA catalysts". Mobile DNA. 5 (1): 8. doi:10.1186/1759-8753-5-8. PMC 3984707. PMID 24612670.
- Peters, Joseph E.; Makarova, Kira S.; Shmakov, Sergey; Koonin, Eugene V. (2017-08-29). "Recruitment of CRISPR-Cas systems by Tn7-like transposons". Proceedings of the National Academy of Sciences. 114 (35): E7358–E7366. doi:10.1073/pnas.1709035114. PMC 5584455. PMID 28811374.
- Tazzyman, Samuel J.; Bonhoeffer, Sebastian (2013). "Fixation probability of mobile genetic elements such as plasmids". Theoretical Population Biology. 90: 49–55. doi:10.1016/j.tpb.2013.09.012. PMID 24080312.
- De Cecco, Marco; Criscione, Steven W.; Peterson, Abigail L.; Neretti, Nicola; Sedivy, John M.; Kreiling, Jill A. (2013). "Transposable elements become active and mobile in the genomes of aging mammalian somatic tissues". Aging. 5 (12): 867–883. doi:10.18632/aging.100621. PMC 3883704. PMID 24323947.
- Antony, Joseph M; Marle, Guido van; Opii, Wycliffe; Butterfield, D Allan; Mallet, François; Yong, Voon Wee; Wallace, John L; Deacon, Robert M; Warren, Kenneth (October 2004). "Human endogenous retrovirus glycoprotein–mediated induction of redox reactants causes oligodendrocyte death and demyelination". Nature Neuroscience. 7 (10): 1088–1095. doi:10.1038/nn1319. PMID 15452578.
- "The Nobel Prize in Physiology or Medicine 1983". nobelprize.org. Retrieved 14 July 2010.
- Keen, E. C. (December 2012). "Paradigms of pathogenesis: Targeting the mobile genetic elements of disease". Frontiers in Cellular and Infection Microbiology. 2: 161. doi:10.3389/fcimb.2012.00161. PMC 3522046. PMID 23248780.
Bibliography
- Miller, W. J.; Capy, P., eds. (2004), Mobile genetic elements: protocols and genomic applications, Humana Press, ISBN 978-1-58829-007-6
- Shapiro, J.A., ed. (1983), Mobile genetic elements, Academic Press, ISBN 978-0-12-638680-6