Gold(I) bromide
Gold(I) bromide can be formed by synthesis from the elements or partial decomposition of gold(III) bromide by careful control of temperatures and pressures. It occurs in two modifications. One (I-AuBr) is isostructural with gold(I) chloride and has a body centered tetragonal unit cell with a=6.734A and c=8.674A and space group I41/amd. The other is isostructural with gold(I) iodide and has a primitive tetragonal cell a=4.296A and c=12.146A and space group P42/ncm. Single crystals of both modifications have been grown by chemical vapor transport.[1] Small amounts of Al,Ga or Fe were used as catalysts for the transport process to obtain the I-AuBr modification.
 | |
| Names | |
|---|---|
| IUPAC name
gold(I) bromide | |
| Other names
Aurous bromide Gold monobromide Gold(I) bromide | |
| Identifiers | |
3D model (JSmol) |
|
| ChemSpider | |
| EC Number |
|
PubChem CID |
|
CompTox Dashboard (EPA) |
|
| |
| Properties | |
| AuBr | |
| Molar mass | 276.87 g/mol |
| Appearance | lemon yellow crystals (AuBr-I), yellow-brown (AuBr-P) |
| Density | 7.9 g/cm3 |
| Melting point | 165 °C (329 °F; 438 K) (decomposes) |
| −61.0·10−6 cm3/mol | |
| Hazards | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa). | |
| Infobox references | |
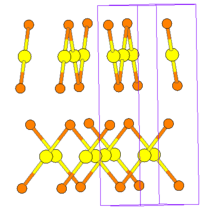
The two structures both consist of -Br-Au-Br-Au-Br- polymeric zigzag chains, but they are stacked in a different arrangement. In the primitive tetragonal phase the chains form layers (see figure) in contrast to the body centered one, where they are more interwoven. Another difference is that the Au-Br-Au angle is only 77° in the former, but 92.3° in the latter.
Density functional calculations on the monohalides of group 11 (Cu, Ag, Au)[2] have tried to shed light on the question why gold halides form rather different, low symmetry structures rather than the cubic zinc blend or rock salt structures of the silver and copper halides. It was shown that this calculation technique accurately predicts which structure type should be stable. The peculiar structures of the gold halides are mostly a result of the relativistic effects that occur for the elements of the later periods of the periodic table.
References
- E.M.W. Janssen and G.A. Wiegers (1978). "Crystal growth and the crystal structures of two modifications of gold monobromide, I-AuBr and P-AuBr". Journal of the Less-Common Metals. 57 (2): P47–P57. doi:10.1016/0022-5088(78)90248-5.
- Tilo Söhnel, Holger Hermann and Peter Schwerdtfeger (2005). "Solid state density functional calculations for the group 11 monohalides". J. Phys. Chem. B. 109 (1): 526–531. doi:10.1021/jp046085y. PMID 16851044.
